10.2(3) Halogenoalkanes & benzene
.png)

 Learning outcomes
Learning outcomes
.png) After studying this sub-topic you should be able to:
After studying this sub-topic you should be able to:
Understand:
- Halogenoalkanes are more reactive than alkanes.
- Halogenoalkanes can undergo nucleophilic substitution reactions.
- Nucleophiles are electron-rich species containing a non-bonding pair of electrons that is donated to an electron-deficient carbon.
- Benzene does not readily undergo addition reactions but does undergo electrophilic substitution reactions.
Apply your knowledge to:
- Write equations for the substitution reactions of halogenoalkanes with aqueous sodium hydroxide.
Relationships & vocabulary
Nature of science
Organic chemical reactions involving functional group interconversions are among the key factors responsible for the progress made in the development and applications of scientific research.
International-mindedness
It is perhaps worth mentioning that benzene is one chemical that is banned for use in school laboratories in almost every country in the world.
For more examples and links to International mindedness, Theory of knowledge, utilization etc. see separate page which covers all of Topics 10 & 20 : Organic chemistry.
Vocabulary
| nucleophile | nucleophilic substitution | electrophile | electrophilic substitution | heterolytic fission |
Learning slides
You can use this slide gallery for learning or for reviewing concepts and information. It covers all the key points in the syllabus for this sub-topic.
Something to think about
The syllabus states:
“Halogenoalkanes are more reactive than alkanes.”
A rather nice Nature of Science exercise might be to question the validity of this statement. Is it a valid statement that is always true?
It is certainly true that alkanes undergo very few different types of chemical reactions. This is mainly due to the relative strengths of the C−H and C−C bonds and the fact that carbon cannot expand its octet. Contrast this with many halogenoalkanes where the carbon atom bonded to the halogen atom is electropositive and can undergo substitution reactions with nucleophiles such as the hydroxide ion, the cyanide ion or ammonia. The statement made in the IB Guide clearly does have some validity. However it is certainly not always the case. One of the hazards associated with aircraft and other vehicles that use alkanes as a fuel is that if there is a fuel leak due to an accident then there is a very real danger of fire. All alkanes are very reactive when it comes to burning them in oxygen. Compare this with halogenoalkanes, in particular fluorocarbons. Fluorine is a highly electronegative element which will make the carbon atom attached to it very electropositive so fluorocarbons would be expected to react readily with nucleophiles. In fact they are completely inert. This is due to the very strong C−F bond. The irony is that certain halogenoalkanes (e.g. bromochlorodifluoromethane, known as halon 1211) are so unreactive that they are carried in aircraft to combat alkane fires – in this instance alkanes are very definitely much more reactive than halogenoalkanes.
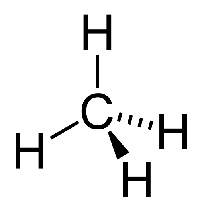
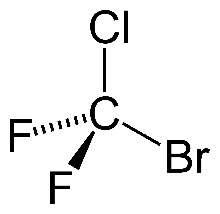 When it comes to combustion, halon1211 (left), a halogenoalkane, is much less reactive than methane (right), an alkane.
When it comes to combustion, halon1211 (left), a halogenoalkane, is much less reactive than methane (right), an alkane.
Another good example to show that in many cases halogenoalkanes are actually less reactive than alkanes is to compare the polymers poly(tetrafluoroethene), PTFE, and poly(ethene). PTFE, which is also known as Teflon®, is the 'non-stick' polymer which is so inert that it is used to coat cooking utensils. According to Plastech the only chemicals known to affect Teflon® finishes are certain alkali metals and some of the most highly reactive fluorinating agents such as xenon difluoride and cobalt(III) fluoride. Compared to this poly(ethene) is much more reactive.
Test your understanding of this topic
(Note that your teacher may have restricted your access to some or all of these questions and worked answers if they are going to use them as a class test or set them as an assignment.)
For ten 'quiz' multiple choice questions with the answers explained see MC test: Halogenoalkanes & benzene.
For short-answer questions see Halogenoalkanes & benzene questions.
More resources
1. One of the problems with videos about nucleophilic substitution is that many of them also go into the SN1 and SN2 mechanisms which are not on the core part of the syllabus. This one, by Master Organic Chemistry, just covers the basics of what a nucleophilic substitution reaction is.
2. There is a similar problem with videos about the structure of benzene at this level as most of them go into the delocalization of pi electrons which is not on the core part of the programme. This one by Allery tutors does cover some of the physical evidence that shows that benzene does not simply consist of three alternate C=C double bonds in a six-membered ring.

 IB Docs (2) Team
IB Docs (2) Team 










