A modern paradigm
 Introduction
Introduction

 The terms paradigm and paradigm shift were first used to describe the way science works by Thomas Kuhn (image on right) in a book called The Structure of Scientific Revolutions published in 1962. Thomas Kuhn (1922-1996) was an American physicist. He contended that science operates in paradigms. A paradigm is an existing framework of knowledge to which all scientists working in the field subscribe to. Paradigms work in various stages.
The terms paradigm and paradigm shift were first used to describe the way science works by Thomas Kuhn (image on right) in a book called The Structure of Scientific Revolutions published in 1962. Thomas Kuhn (1922-1996) was an American physicist. He contended that science operates in paradigms. A paradigm is an existing framework of knowledge to which all scientists working in the field subscribe to. Paradigms work in various stages.The phases of a scientific paradigm
1. The pre-paradigm stage
During this period there are likely to be several competing schools of thought. For example, is matter continuous or is it made up of discrete individual particles. Each school of thought defends its position by referring to the data or phenomena that best supports its view.
2. The establishment of a paradigm
Eventually one of the pre-paradigm schools of thought will gain the ascendency and will be more successful at explaining phenomena than one of the rival theories. This theory will be accepted by many. It will be able to make successful predictions and will form the basis of research for people working in the field.
3. The working paradigm or ‘normal science’
Scientists begin to answer the questions which are posed by the paradigm. They make ever more sophisticated measurements, publish erudite scientific papers and no longer need to defend their assumptions as the rest of the scientific community accepts the basis of their work.
4. Crisis
Some data or phenomena are found which do not fit the existing paradigm. These failures are tolerated and the paradigm becomes stretched to accommodate them (see phlogiston below).
5. Paradigm shift
Eventually a scientist will propose a new theory which is outside the existing paradigm. This new paradigm not only explains the facts that fitted the old paradigm but also accounts for all (or at least most of) the anomalies found during the crisis stage. Older workers in the field tend to resist the new ideas (as it undermines all their previous work). However if the new theory is promising it will attract supporters and new scientists will be educated using the new paradigm. Older worker who do not change their mind become ignored and eventually die out. A period of normal science again sets in until a new crisis occurs.
An example of a classic paradigm
There have been many classic paradigm shifts in science in the past and the TOK teachers will tend to give these classic examples. They include Darwin’s theory of evolution, Galileo’s theory of a heliocentric universe and Einstein’s replacement of Newtonian mechanics. All scientists are currently working within paradigms and it is difficult to realise that in the future people may look back in amazement at what people in the twenty first century believed. After all it is still less than two hundred years today (pre-Darwin) since people believed Bishop Usher’s calculations that humans have only been on the Earth since 4004 BC when all creation began.
Phlogiston
There is one classic chemistry paradigm which some non-chemistry TOK teachers may also use in their classes – the paradigm of phlogiston. Phlogiston theory was the first unifying theory of Chemistry. In 1669 Johann Becher (1635-1682) maintained that all bodies are composed of air, water and the three earths – terra pinguis (fatty earth), terra mercurialis (mercurial earth) and terra lapidea (stony earth). It was Georg Stahl (1660-1734) who later replaced terra pinguis with phlogiston (which means burnt). Stahl defined combustion as the loss of phlogiston. This fitted many observed facts such as when a piece of wood burns it leaves behind only a small amount of ash. The paradigm explains this by stating that when the wood burns it gives off the phlogiston to the surroundings as heat and light. For about 100 years this paradigm was accepted by scientists as an explanation for combustion. Two facts illustrate how scientists become trapped within the accepted paradigm. When the mass of a metal was weighed after it had combusted the residue (metal oxide) weighed more than the unburned metal. The only possible explanation within the paradigm is that phlogiston can have negative mass. Oxygen was discovered in 1774 by Joseph Priestley (1733-1804). However Priestly was such an adherent of phlogiston theory that he failed to realise the significance of his discovery and called his new gas ‘dephlogisticated air’ to explain why substances burn well in it.

Antoine Lavoisier (with his wife Marie Paulze) painted by Jacques Louis David in 1788. This painting hangs in the Science Museum in London where I took this photograph
The paradigm shift came from Antoine Lavoisier (1743-1794). He made quantitative measurements of the reaction of mercury with oxygen and showed conclusively in 1777 that combustion involved the addition of oxygen.
An example of a more modern chemistry paradigm
What you can do in your classes is illustrate a more modern paradigm shift that has occurred in Chemistry as you cover covalent bonding. This can then provide students with a useful example to possibly include in their TOK essay as well as encourage them to think more critically about chemistry. So here is the ‘modern’ paradigm.
Most teachers introduce covalent bonding by looking at how two hydrogen atoms combine.
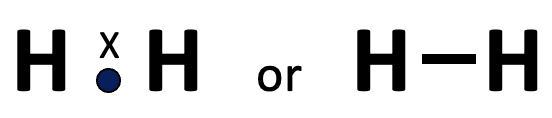
This leads to the definition of covalent bonding which forms the basis of the paradigm of Gilbert Lewis (1875-1946).
When two atoms form a covalent bond each contributes one electron so that each atom then has a share in a noble gas configuration.
If this is extended to fluorine and chlorine and compounds such as water and methane the paradigm works well.
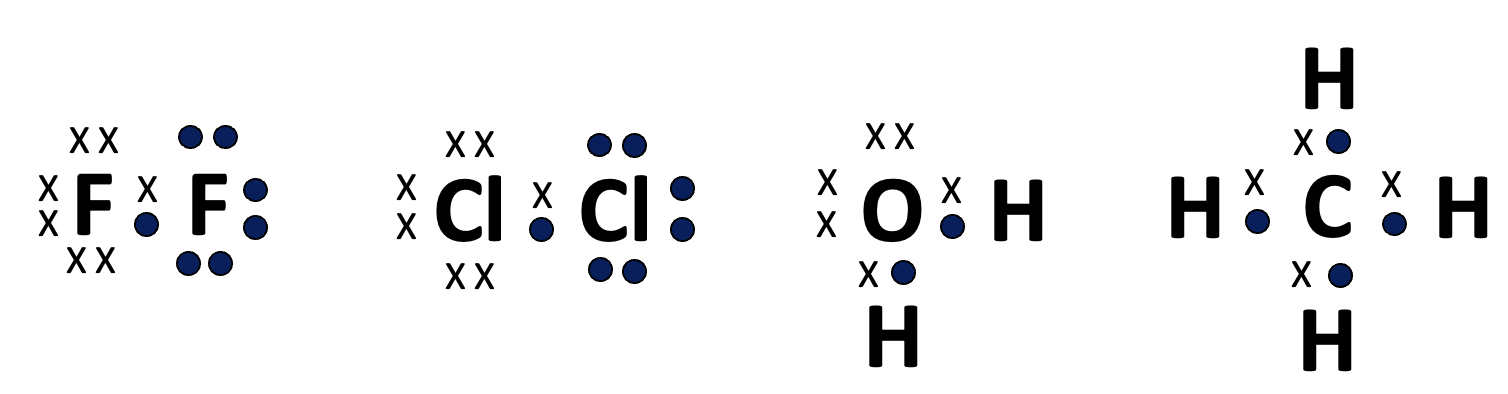
The paradigm needs to be altered slightly so that atoms can share more than one electron from each atom to form double and triple bonds in substances like oxygen and nitrogen respectively.
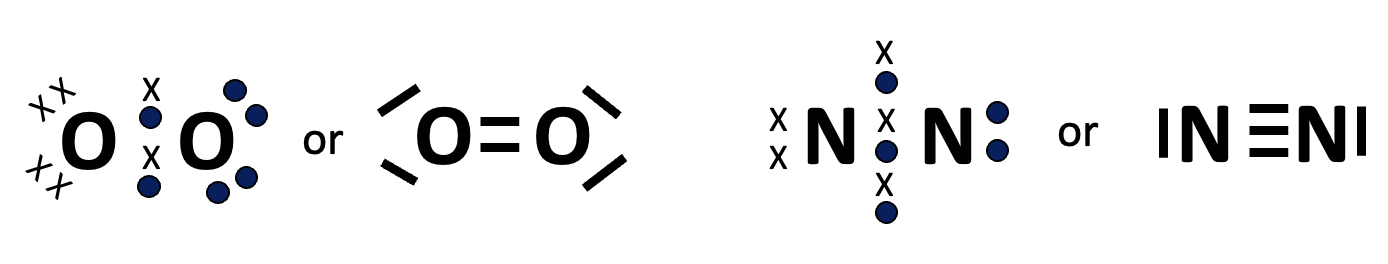
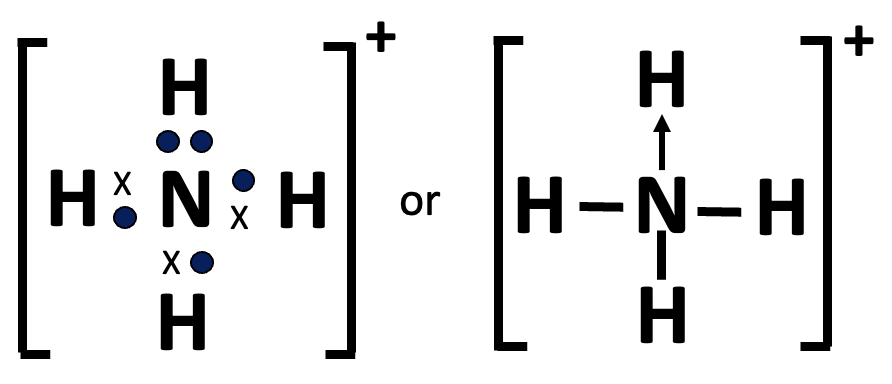
It needs to be altered again to account for carbon monoxide or the ammonium ion. Now one of the atoms can provide both of the electrons in the shared pair (this is known as dative or coordination covalent bonding).
.png)
The paradigm needs to be stretched further to accommodate compounds like sulfur dioxide and ozone and the concept of resonance hybrid needs to be included in the paradigm.


To account for compounds like phosphorus(V) pentachloride and sulfur(VI) hexafluoride the paradigm needs to accommodate the fact that the atoms can ‘expand their octet’ and that the outer electron pairs do not have to make a noble gas configuration.
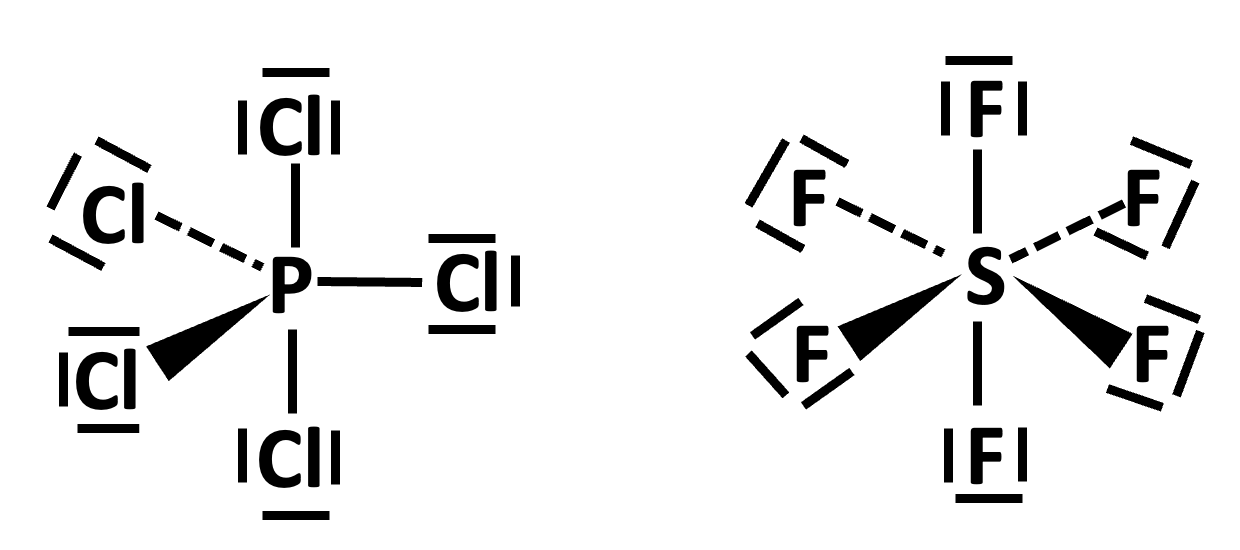
Similarly the paradigm also needs to be modified to account for electron deficient compounds such as boron trifluoride and beryllium dichloride which contain only six and four electrons respectively around the central atom (and hence act as good Lewis acids).
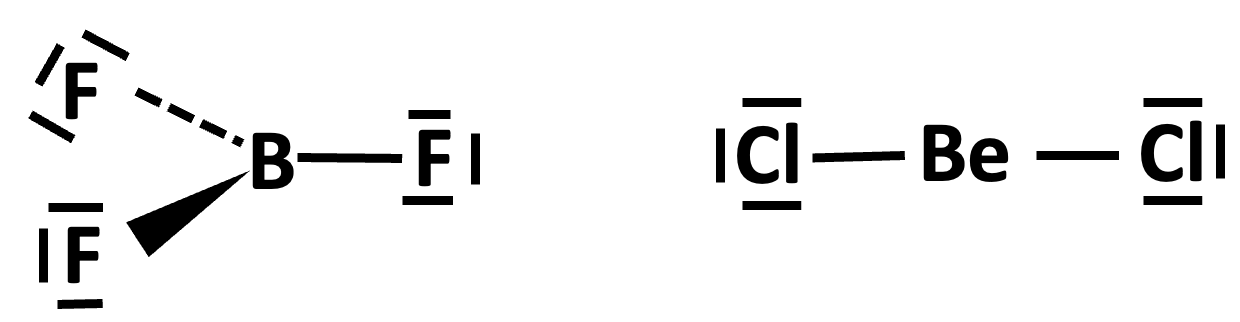
We have come a long way from the initial paradigm but the Lewis Structure paradigm of covalent bonding still just about works with a huge number of modifications. The crisis arises with nitrogen(II) oxide, NO. This has an odd number of electrons and simply does not fit the paradigm. With the benefit of hindsight Higher Level students can already spot one wrong assumption in the Lewis Structure paradigm – all electrons are being treated as equal whereas they occupy different sub-orbitals (s, p and d etc.) and do not all have the same energy.
The paradigm shift comes with a completely new theory – in fact two closely related theories - valence bond theory and molecular orbital theory. Molecular orbital theory looks at the linear combination of atomic orbitals to form both bonding and anti-bonding molecular orbitals and neatly explains the bonding in nitrogen(II) oxide. It is beyond IB level, but one of its strong points as a ‘new paradigm’ is that not only does it explain nitrogen(II) oxide but it also explains why oxygen is paramagnetic. Under Lewis theory oxygen contains two shared pairs of electrons making up the double bond which would make it diamagnetic but in molecular orbital theory oxygen molecules have two unpaired electrons which account for its paramagnetic properties.

 IB Docs (2) Team
IB Docs (2) Team