Environmental impact - plastics

 A.7 Environmental impact - plastics (2 hours)
A.7 Environmental impact - plastics (2 hours)
Pause for thought
Three highly relevant items for ‘Pause for thought’ have already been included elsewhere. The harmful effects of low molecular mass phthalate esters are discussed in A.5 Polymers, the effects of PCBs on the environment are discussed in B.6 Biochemistry & the environment and the effects of the dioxins used in the Vietnam War are discussed in the practical on the Hydrolysis of halogenoalkanes.
This ‘Pause for thought’ focuses on the Great Pacific Garbage Patch, which is alluded to under International Mindedness. A quick search will reveal many articles written on this. One good one is by Ocean CleIB Docs (2) Teamp. Essentially the Great Pacific Garbage Patch consists of not one but two separate patches, one located off the coast of Japan and the other between Hawaii and California.
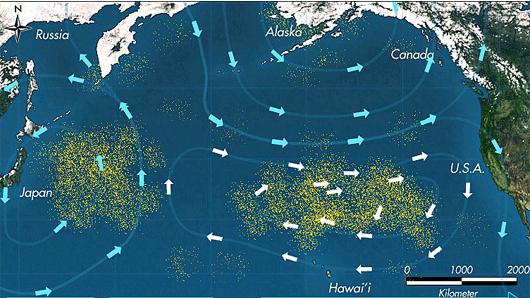
The two 'Garbage Patches' in the Pacific Ocean - image from sdca.wordpress.com
They are caused by ocean gyres. A gyre describes the system of circular currents in the ocean caused by a combination of the Earth’s wind patterns and forces due to the Earth’s rotation. In the centre of these gyres the sea is relatively calm so all the debris collects there. Most of this is plastic due to the fact that most plastics do not degrade at all readily in the environment. You can find many videos on this but one of the comments often attached to them is “if this garbage patch is so big why do you not actually show pictures of it?” The reason is that although the plastics do not decompose chemically they do break down physically into ever more smaller particles known as ‘microplastics’ many of which are too small to be seen with the naked eye. The videos tend to concentrate on the larger pieces of plastic found in the stomach of birds etc. but the microplastics are also extremely harmful particularly to plankton feeders. They also reduce the amount of sunlight penetrating below the ocean’s surface. Because the plastics ‘belong’ to no one country and the cost of cleaning up the ocean would be huge, currently very little is being done to solve the problem. The effect on marine wildlife is huge and the problem is continually increasing even though eventually some of the plastic does sink to the ocean floor.
Nature of Science
The development and use of plastics is a good example of how scientific research often proceeds with perceived benefits in mind. Only later do the risks and implications become more apparent.
Learning outcomesAfter studying this topic students should be able to: Understand:
Apply their knowledge to:
| Clarification notesDioxins have a very long half-life in the environment and can be passed on in the food chain. Polychlorinated dibenzodioxins (PCDDs) and PCBs should both be considered as examples of carcinogenic chlorinated dioxins or dioxin-like substances. Phthalate esters should be considered as examples of plasticizers. Many toxins can be released in house fires due to the combustion of plastics (shower curtains, etc.). To help prevent these hazards low smoke zero halogen cabling is often used in wiring. Resin Identification Codes (RICs) can be found in Section 30 of the data booklet. The structures of various materials molecules are given in Section 31 of the data booklet. International-mindednessAlthough a Mobius strip, designed in 1960s, is used as the international symbol for recycle, reuse and reduce, global recognition of this symbol ranks well below other symbols. What factors influence the recognition of symbols?How can the problem of plastic floating in the Pacific Ocean gyre (known as 'the Great Pacific Garbage Patch' or 'Pacific trash vortex') be addressed internationally? |
Teaching tipsAlthough the heading of this topic is “Environmental impact – plastics” it is important to impress upon students that it does not only consider plastics. Chemicals used in their production (e.g. phthalate esters used as plasticizers) or compounds formed when certain plastics combust (e.g. dioxins) are also included in this topic. PCBs actually have no clear link with plastics, but are included because they share a structural similarity and toxicity to dioxins and their use has resulted in man-made pollution of the environment. Even though the use of PCBs was banned in 1970 their effects are still prevalent today. Students should be able to recognise the basic structures of dioxins, PCBs, PCDDs and possibly phthalate esters. They should also be able to recognise absorptions due to specific functional groups in the infrared spectra of a compound in order to identify plastic types when using the Resin Identification Code (RIC). For example, polyethylene terephthalate, PET or PETE, shows a strong absorption at 1700-1750 cm-1 due to the C=O group and PVC shows an absorption between 600-800 cm-1 due to the C-Cl bond. Students are also expected to write equations for the combustion of polymers although these are presumably simple equations and would not include the formation of dioxins. Other than these structures, i.r. absorptions and combustion equations the remainder of this topic is quite factual. Students will need to be aware of the fact that polymers are generally unreactive due to the strong covalent bonds they contain (although the C-Cl bond in chlorinated polymers is not so strong), this mainly explains why they persist in the environment. They also need to know the sources of the compounds mentioned and their health and environmental implications. Stress too the importance of recycling wherever possible and the need for high temperature incinerators when plastics are disposed of by combustion to ensure no dioxins are formed. Due to this and other reasons the disposal and recycling of plastics is an energy intensive process. For some reason biodegradable plastics are not included in the programme so do not need to be covered, although they are probably worth mentioning in passing. | Study guide
Page 118 QuestionsFor ten 'quiz' questions (for quick testing of knowledge and understanding with the answers explained) see MC test: Environmental impact - plastics. For short-answer questions see Environmental impact - plastics questions together with the worked answers on a separate page Environmental impact - plastics answers. Vocabulary listdioxin |
Teaching slides
Teachers may wish to share these slides with students for learning or for reviewing key concepts.
Other resources
1. Dioxins, what they are and their possible health effects by IAQ Video Network.
![]() Dioxins and their health effects
Dioxins and their health effects
2. A sobering account of the effects of Agent Orange 35 years after the end of the Vietnam War.
![]() The after effects of Agent Orange
The after effects of Agent Orange
3. PCBs, what they are, where they are found and the problems they cause by Paul Cochrane.
4. Boyan Slat proposes a radical clean-up solution to the Great Pacific Garbage Patch problem.
![]() Solving the Great Pacific Garbage Patch problem
Solving the Great Pacific Garbage Patch problem
5. A good resource for the infrared spectra of various plastics can be found at the site by DoITPoMS from the University of Cambridge. One of the most obvious things it shows is the difference between PTFE and all the other plastics as there is no absorption at about 3000 cm-1 in the IR spectrum, since PTFE contains no C-H bonds.

 IB Docs (2) Team
IB Docs (2) Team 



















