Synthetic routes

 20.2 Synthetic routes (2 hours)
20.2 Synthetic routes (2 hours)
Pause for thought

Which route to take?
This a rather strange sub-topic as basically it just makes links between other organic sub-topics and the reactions contained in them which is what a good teacher (and student) should be doing anyway! It does limit the number of stages to four which is ample as there are not actually that many reactions that are covered anyway. It can be useful to list these reactions:

What is significant, although perhaps not immediately obvious, is that many of these reactions only go one way. For example, alkenes react with hydrogen halides to give halogenoalkanes but the elimination of hydrogen halides from halogenoalkanes to give alkenes is not included in the syllabus. Similarly, esterification is on the core/AHL programme but saponification, the reverse process, is not. For this reason it is perhaps easier to show the possible reactions in a flow diagram:
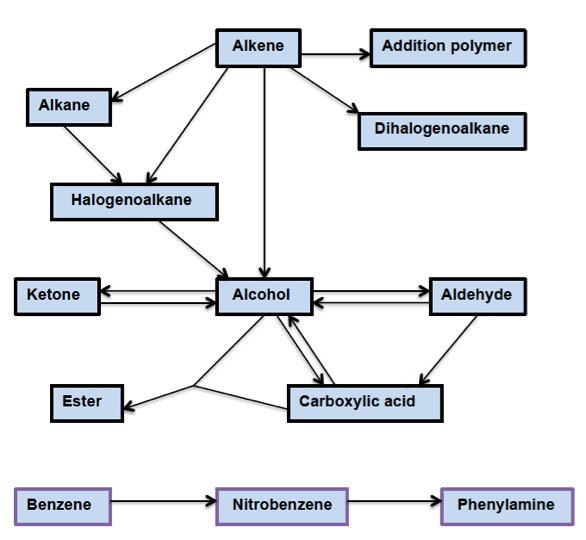
An analysis of this diagram does show that the range of syntheses that can be asked is quite limited. For example, the only synthesis involving arenes is the conversion of benzene into phenylamine via nitrobenzene and the route to an aldehyde, ketone or carboxylic acid is likely to come via the conversion of an alkene or halogenoalkane to an alcohol.
Nature of Science
The thinking processes of organic chemists when designing syntheses can also involve retro-synthesis and the ability and imagination to think in a reverse-like manner. This is an important facet of the scientific method.
Learning outcomesAfter studying this sub-topic students should be able to: Understand:
Apply their knowledge to:
| Clarification notesOnly conversions involving four or less discrete steps will be assessed in synthetic routes. Any of the reaction types covered in Topic 10 and sub-topic 20.1 can be used. International-mindednessConsider the importance of natural products (and the compounds that can be synthesised from them) to developing countries and their wider importance to the developed world. |
Teaching tipsI think a good teacher would already have been making these connections as they taught all the previous sub-topics so in a sense no extra teaching is needed. In fact this sub-topic is a good way to revise/review the organic reactions already covered. I do spend just a little time explaining the importance of reaction pathways in, for example, the organic synthesis of new drugs. It is worth stressing the importance of obtaining a high yield for each step since in a multi-step synthesis the final yield (based on the starting material) may be very low. It might be worth giving the example of Bob Woodward (see below) whose 55 separate step synthesis of chlorophyll in 1960 earned him the Nobel prize. It is worth showing one or two examples to students and stress that often it worth considering working backwards, i.e. looking at the product and seeing what could be reacted to form it - the basis of retro-synthesis. Stress that the key is the conversion of functional groups into different functional groups. It is also worth pointing out that often there may be more than one way of achieving the synthesis. For example, an alcohol can be synthesised directly from an alkene by hydration or by first adding hydrogen bromide to give a bromoalkane that can then undergo nucleophilic substitution with warm dilute aqueous sodium hydroxide solution. It might also be worth emphasising the importance of Markovnikov's rule to give a desired product when halogenoalkanes are added to asymmetric alkenes. What is required is for students to get practice at designing these reaction pathways as it is also a very useful way for them to learn the required organic chemistry together with the necessary conditions required for the reaction to proceed. | Study guide
Page 93 QuestionsFor ten 'quiz' multiple choice questions with the answers explained see MC test: Synthetic routes. For short-answer questions which can be set as an assignment for a test, homework or given for self study together with model answers see Synthetic routes questions. Vocabulary listretro-synthesis IM, TOK, Utilization etc.See separate page which covers all of Topics 10 & 20. |
Teaching slides
Teachers may wish to share these slides with students for learning or for reviewing key concepts.
Other resources
1. The separate reactions have already been covered in other sub-topics. Out of interest you may like to impress upon students the relevance of reaction pathways by showing them how Bob Woodward achieved his multistage synthesis of chlorophyll in 1960. The full paper showing all 55 steps is surely one of the greatest of all chemistry research papers ever to be produced. A shortened version of the pathways for chlorophyll A is also available.
2. The problem with most other video resources on synthesis and retro-synthesis is that they use examples that involve many more reactions (e.g. elimination, Friedel Craft and Grignard reactions etc.) that are not covered on the IB programme so are not particularly helpful to IB students. This is also even true for videos on this topic on the past IB programme as the list of reactions has changed considerably and in the last programme the pathways were limited to just two steps. One set of notes that may be of some help is from A-levelchemistry which has been compiled by Gordon Watson from Kelso High School.

 IB Docs (2) Team
IB Docs (2) Team 

















