5. Energetics & thermochemistry
 Energetics / thermochemistry
Energetics / thermochemistry
For each question choose the answer you consider to be the best.
1. Which is an exothermic process?
A. K(s) → K(g)
B. H2O(l) → H+(aq) + OH–(aq)
C. Cl(g) + e– → Cl–(g)
D. Mg(g) → Mg+(g) + e–
2. When some solid ammonium nitrate was dissolved in water the temperature decreased
from 22 oC to 3 oC. What can be deduced from this observation?
A. The dissolving is endothermic and ∆H is positive.
B. The dissolving is endothermic and ∆H is negative.
C. The dissolving is exothermic and ∆H is positive.
D. The dissolving is exothermic and ∆H is negative.
3. Which is a correct statement about an endothermic reaction?
A. The bonds in the reactants are stronger than in the products and ∆H is positive.
B. The bonds in the products are stronger than in the reactants and ∆H is positive.
C. The bonds in the reactants are stronger than in the products and ∆H is negative
D. The bonds in the products are stronger than in the reactants and ∆H is negative.
4. Which of the following conditions commonly apply to the standard enthalpy change for a reaction, ∆H?
I. A pressure of 100 kPa
II. A temperature of 298 K
III. One mol of all reactants and all products
A. I and II only
B. I and III only
C. II and III only
D. I, II and III
5. Which is a correct statement about the following enthalpy level diagram for a reaction?

A. The reaction is exothermic and ∆H is positive.
B. The reaction is exothermic and ∆H is negative.
C. The reaction is endothermic and ∆H is positive.
D. The reaction is endothermic and ∆H is negative.
6. Which statements are correct for all exothermic reactions?
I. The products are more stable than the reactants
II. The bonds in the products are stronger than the bonds in the reactants
III. The enthalpy of the products is less than the enthalpy of the reactants
A. I and II only
B. I and III only
C. II and III only
D. I, II and III
7. For which of the following is the sign of the enthalpy change different to the other three?
A. Br2(g) → 2Br(g)
B. Br(g) → Br+(g) + e–
C. Br(g) + e– → Br–(g)
D. Br(l) → Br(g)
8. Which is a correct statement concerning the reaction shown?
N2(g) + 3H2(g) → 2NH3(g) ∆H![]() = – 92 kJ
= – 92 kJ
A. 92 kJ of energy are absorbed for every mol of ammonia formed.
B. 92 kJ of energy are released for every mol of ammonia formed.
C. 46 kJ of energy are absorbed for every mol of ammonia formed.
D. 46 kJ of energy are released for every mol of ammonia formed.
9. The heat capacity of manganese is 0.480 kJ kg-1 K-1. How much energy is required to raise
the temperature of 3.00 kg of manganese by 20.0 K?
A. 28.8 kJ
B. 3.20 kJ
C. 0.0720 kJ
D. 288 kJ
10. The specific heat capacities (in kJ kg-1 K-1) of four elements are:
carbon (graphite) 0.709
copper 0.385
gold 0.129
iron 0.449
Which element will show the greatest increase in temperature if 2 kJ of heat is supplied to 0.100 kg samples of each element at the same initial temperature?
A. carbon (graphite)
B. copper
C. gold
D. iron
11. When 25 cm3 of 1.0 mol dm-3 HCl was added to 25 cm3 of 1.0 mol dm-3 KOH the temperature
increased by 7 oC.
Which combination of HCl and KOH will also give a temperature rise of 7 oC?
A. 50 cm3 of 0.25 mol dm-3 HCl and 50 cm3 of 0.25 mol dm-3 KOH.
B. 50 cm3 of 0. 50 mol dm-3 HCl and 50 cm3 of 0.50 mol dm-3 KOH.
C. 50 cm3 of 1.0 mol dm-3 HCl and 50 cm3 of 1.0 mol dm-3 KOH.
D. 50 cm3 of 2.0 mol dm-3 HCl and 50 cm3 of 2.0 mol dm-3 KOH.
12. The heat produced when 0.010 mol of propanone was combusted raised the temperature
of 250 g of water by 14 oC.
The specific heat capacity of water is 4.2 J g-1 K-1.
Which is the correct expression for value of the enthalpy of combustion of propanone (in J mol-1)?
A. − 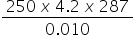
B. − 
C. − ![]()
D. − (250 x 4.2 x 14 x 0.010)
13. How much energy is released when 14.0 g of carbon monoxide are completely combusted?
2CO(g) + O2(g) → 2CO2(g) ∆H = – 560 kJ
A. 1120 kJ
B. 560 kJ
C. 280 kJ
D. 140 kJ
14. When one mol of magnesium and one mol of hydrogen are combusted the enthalpy changes are - 602 kJ
and - 242 kJ respectively.
Mg(s) + ½ O2(g) → MgO(s) ∆H = – 602 kJ
H2(g) + ½ O2(g) → H2O(g) ∆H = – 242 kJ
What is the enthalpy change for the reduction of one mol of magnesium oxide by hydrogen?
MgO(s) + H2(g) → Mg(s) + H2O(g)
A. + 844 kJ
B. + 360 kJ
C. – 360 kJ
D. – 844 kJ
15. The enthalpy change for the dimerisation of two mol of nitrogen dioxide to form one
mol of dinitrogen tetroxide is – 57 kJ.
2NO2(g) → N2O4 ∆H![]() = – 57 kJ
= – 57 kJ
The enthalpy change for the formation of one mol of NO2(g) from its elements is + 33 kJ
½ N2(g) + O2(g) → NO2(g) ∆H![]() = +33 kJ
= +33 kJ
What is the enthalpy change for the formation of one mol of dinitrogen tetroxide from its elements?
N2(g) + 2O2(g) → N2O4(g)
A. + 9 kJ
B. – 9 kJ
C. + 24 kJ
D. – 24 kJ
16. The enthalpy changes for the formation of one mol of each of the two oxides of phosphorus
from their elements are given below:
P4(s) + 3O2(g) → P4O6(s) ∆H![]() = – 1600 kJ
= – 1600 kJ
P4(s) + 5O2(g) → P4O10(s) ∆H![]() = – 3000 kJ
= – 3000 kJ
What is the enthalpy change for the oxidation of one mol of P4O6 to form P4O10?
P4O6(s) + 2O2(g) → P4O10(s)
A. – 4600 kJ
B. + 4600 kJ
C. + 1400 kJ
D. – 1400 kJ
17. Consider the following three bond enthalpies (in kJ mol-1)
H—H 436
Cl—Cl 243
H—Cl 432
What is the enthalpy change (in kJ) for the reaction below?
2HCl(g) → H2(g) + Cl2(g)
A. + 185
B. + 237
C. – 185
D. – 237
18. Which of the following enthalpy changes can be calculated using only bond enthalpies
and average bond enthalpies?
I. C2H4(g) + H2(g) → C2H6(g)
II. C8H16(l) + H2(g) → C8H18(l)
III. CH4(g) + 4F2(g) → CF4(g) + 4HF(g)
A. I and II only
B. I and III only
C. II and III only
D. I, II and III
19. The bond enthalpies for N≡N and H—H are 945 kJ mol-1 and 436 kJ mol-1 respectively.
When nitrogen and hydrogen react to form ammonia the enthalpy change is – 92 kJ.
N2(g) + 3H2(g) → 2NH3(g) ∆H![]() = – 92 kJ
= – 92 kJ
What is the average bond enthalpy (in kJ) for the N—H bond?
A. 212
B. 424
C. 391
D. 782
20. Ethane can react with chlorine in ultraviolet light to form chloroethane:
C2H6(g) + Cl2(g) → C2H5Cl(g) + HCl(g)
The relevant bond enthalpies are

What is the enthalpy change, ∆H![]() , for this reaction (in kJ)?
, for this reaction (in kJ)?
A. – 99
B. + 99
C. + 341
D. – 315
 Answers
Answers
1. C, 2. A, 3. A, 4. A, 5. B, 6. D, 7. C, 8. D, 9. A, 10. C,
11. C, 12. B, 13. D, 14. B, 15. A, 16. D, 17. A, 18. B, 19. C, 20. A.
Download the Energetics/thermochemistry ![]() Topic 5 MC test
Topic 5 MC test ![]()
Download the answer grid with the correct answers to cut out for Topic 5 MC Answers ![]()

 IB Docs (2) Team
IB Docs (2) Team