Hess's Law

 5.2 Hess's Law (2 hours)
5.2 Hess's Law (2 hours)
Pause for thought
Hess’s Law is named after Germain Henri Hess (1802 – 1850) a Swiss national who moved with his family to Russia at the age of 3. He qualified as a doctor but later became a professor of chemistry at the St Petersburg Technical Institute. He was interested in minerals and analyzed one particular ore of silver telluride, Ag2Te, which is now called hessite in his honour.

Germain Hess Hessite
Hess’s Law states that the total energy change in a chemical reaction depends only upon the initial and final states and is independent of the reaction pathway. It used to be known as the Law of Constant Heat Summation and is in fact another way of stating the Law of Conservation of Energy.
So how can Hess’s Law be applied?
There are basically three different methods that can be used to solve Hess’s Law problems:
- Energy cycle
- Enthalpy level diagram
- Simultaneous equations
So which is the best method to train students to use?
Energy cycle, enthalpy level diagram or simultaneous equations?
Take as an example Question 15 on the SL Energetics/thermochemistry multiple choice test.
Qu.15. The enthalpy change for the dimerization of two mol of nitrogen dioxide to form one mol of dinitrogen tetroxide is – 57 kJ.
2NO2(g) → N2O4(g) ∆H![]() = – 57 kJ
= – 57 kJ
The enthalpy change for the formation of one mol of NO2(g) from its elements is + 33 kJ
½ N2(g) + O2(g) → NO2(g) ∆H![]() = + 33 kJ
= + 33 kJ
What is the enthalpy change for the formation of one mol of dinitrogen tetroxide from its elements?
N2(g) + 2O2(g) → N2O4(g)
1. Solution using simultaneous equations.
To get N2 on the left hand side start with the second equation and double it:
N2(g) + 2O2(g) → 2NO2(g) ∆H![]() = + 66 kJ
= + 66 kJ
To get rid of 2NO2 and to get N2O4 on the right hand side add the first equation:
N2(g) + 2O2(g) → N2O4(g) ∆H![]() = + 66 + (– 57) = + 9 kJ
= + 66 + (– 57) = + 9 kJ
Easy - but it does not really involve any chemistry.
2. Solution using an enthalpy diagram.
The problem is that you need to put the reactants and products in the correct places on the diagram.

Personally I do not find it very obvious from the information given even though there is no need to include the oxygen (as you do not know the answer beforehand) and students certainly do not find this easy.
Once the diagram has been drawn correctly it is easy to see that x is positive and is the difference in value between 66 and 57 kJ.
3. Solution using an energy cycle.
Use the idea of A going directly to B or via C. In this case A is N2, B is N2O4 and C is NO2.
In chemistry terms nitrogen can either be oxidized directly to N2O4 or first to NO2 which then dimerizes
to N2O4. (Again there is no need to include the oxygen in the cycle.)
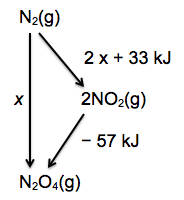
From the diagram (i.e. applying Hess’s Law) it is easy to see that x = (2 x 33) + (– 57) = + 9 kJ
All three methods are correct but I think the energy cycle method is the easiest, the most elegant and demonstrates the best understanding of chemistry.
Nature of Science
Testing hypotheses. Hess's Law states that if the same products are formed from the same initial reactants then the energy change should be the same regardless of the number of steps. This is an example of where a hypothesis based on the law of conservation of energy and on atomic theory can be tested experimentally.
Learning outcomesAfter studying this topic students should be able to: Understand
Apply their knowledge to:
| Clarification notesValues for enthalpies of formation can be found in Section 12 of the data booklet. One application of Hess's Law is ΔH reaction = Σ(ΔHf⦵ International-mindednessThere is a rather strange reference in the syllabus about recycling. It is not clear why this is under a sub-topic on Hess's Law but it states that although recycling materials is often an effective means of reducing the environmental impact of production, its efficiency in energy terms can vary in different countries. |
Teaching tipsI usually introduce this by asking students to write the equation for the formation of methane. If they have understood the definition they should write: C(s) + 2H2(g) → CH4(g) I then ask them how they would determine ΔHf⦵ for the reaction. They often try to devise some sort of apparatus to measure it directly but then realise that there is no way in which they can ensure that the only product of reacting carbon with hydrogen is methane and not some other hydrocarbon. They can however determine directly the enthalpies of combustion of carbon, hydrogen and methane. This leads to the concept of energy cycles and how the enthalpy of formation of methane can be determined indirectly. Although the formal definition of Hess's Law is not required it is important to stress that it is just another way of stating the law of conservation of energy. If Hess's Law is not true then we could use an energy cycle to produce a perpetual motion machine - i.e. solve all the world's energy problems! Stress throughout that the correct states are needed and that if one or more changes of state occur then this too needs to be incorporated into the energy cycle. The topic lends itself to good practical work. Classic experiments are the hydration of copper(II) sulfate (see enthalpy changes right), the enthalpy of formation of metal oxides and the decomposition of potassium hydrogen carbonate. Some practicals available on the web attempt to verify Hess's Law by carrying out three separate reactions. For example, adding NaOH(s) to water, adding NaOH(s) to HCl(aq) and then adding NaOH(aq) to HCl(aq). However this seems to me to miss the point as it is better applied to reactions where the experimental value cannot be determined directly. | Study guide:
Page 40 QuestionsFor ten 'quiz' multiple choice questions with the answers explained see MC test: Hess's Law. For short-answer questions which can be set as an assignment for a test, homework or given for self study together with model answers see Hess's Law questions. Vocabulary listHess's Law IM, TOK, Utilization etc.See separate page which covers all of Topics 5. Practical work |
Teaching slides
Teachers may wish to share these slides with students for learning or for reviewing key concepts.
Other resources
1. A tutorial which explains how to solve Hess's Law problems just by using simultaneous equations. It works, but in my view (see above), by ignoring the use of energy cycles it does not show any real understanding.
2. Students teaching students. It could be worth showing this to students as they might appreciate it. Although quite thorough it does use three separate reactions simply to verify Hess's Law rather than using it to find out a value that cannot be determined directly.

 IB Docs (2) Team
IB Docs (2) Team 









