| Date | May 2018 | Marks available | 2 | Reference code | 18M.2.sl.TZ2.7 |
| Level | SL | Paper | 2 | Time zone | TZ2 |
| Command term | Discuss | Question number | 7 | Adapted from | N/A |
Question
The structure of an organic molecule can help predict the type of reaction it can undergo.
Improvements in instrumentation have made identification of organic compounds routine.
The empirical formula of an unknown compound containing a phenyl group was found to be C4H4O. The molecular ion peak in its mass spectrum appears at m/z = 136.
The Kekulé structure of benzene suggests it should readily undergo addition reactions.
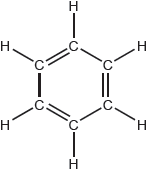
Discuss two pieces of evidence, one physical and one chemical, which suggest this is not the structure of benzene.
Formulate the ionic equation for the oxidation of propan-1-ol to the corresponding aldehyde by acidified dichromate(VI) ions. Use section 24 of the data booklet.
The aldehyde can be further oxidized to a carboxylic acid.
Outline how the experimental procedures differ for the synthesis of the aldehyde and the carboxylic acid.
Deduce the molecular formula of the compound.
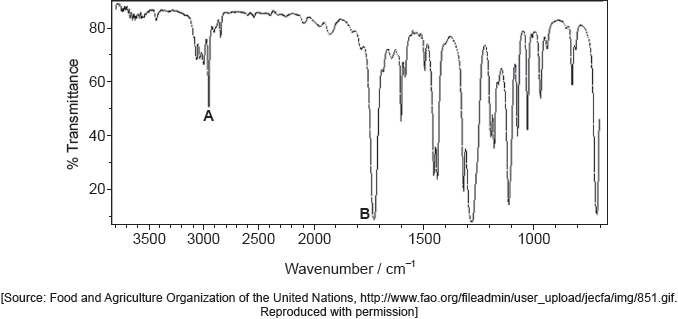
Identify the bonds causing peaks A and B in the IR spectrum of the unknown compound using section 26 of the data booklet.
Deduce full structural formulas of two possible isomers of the unknown compound, both of which are esters.
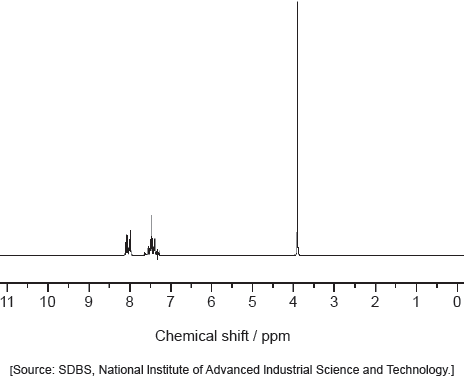
Deduce the formula of the unknown compound based on its 1H NMR spectrum using section 27 of the data booklet.
Markscheme
Physical evidence:
equal C–C bond «lengths/strengths»
OR
regular hexagon
OR
«all» C–C have bond order of 1.5
OR
«all» C–C intermediate between single and double bonds
Chemical evidence:
undergoes substitution reaction «more readily than addition»
OR
does not discolour/react with bromine water
OR
substitution forms only one isomer for 1,2-disubstitution «presence of alternate double bonds would form two isomers»
OR
more stable than expected «compared to hypothetical molecule cyclohexa-1,3,5-triene»
OR
enthalpy change of hydrogenation/combustion is less exothermic than predicted «for cyclohexa-1,3,5-triene»
M1:
Accept “all C–C–C bond angles are equal”.
[2 marks]
3CH3CH2CH2OH(l) + Cr2O72–(aq) + 8H+(aq) → 3CH3CH2CHO(aq) + 2Cr3+(aq) + 7H2O(l)
correct reactants and products
balanced equation
[2 marks]
Aldehyde:
by distillation «removed from reaction mixture as soon as formed»
Carboxylic acid:
«heat mixture under» reflux «to achieve complete oxidation to –COOH»
Accept clear diagrams or descriptions of the processes.
[2 marks]
«\(\frac{{136}}{{48 + 4 + 16}} = 2\)»
C8H8O2
[1 mark]
A: C–H «in alkanes, alkenes, arenes»
AND
B: C=O «in aldehydes, ketones, carboxylic acids and esters»
[1 mark]
Any two of:
OR C6H5COOCH3
OR CH3COOC6H5
OR HCOOCH2C6H5
Do not penalize use of Kekule structures for the phenyl group.
Accept the following structures:
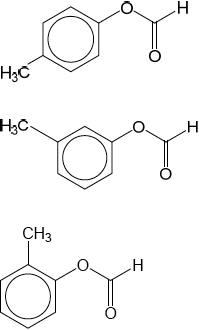
Award [1 max] for two correct aliphatic/linear esters with the molecular formula C8H8O2.
[2 marks]
C6H5COOCH3 «signal at 4 ppm (3.7 – 4.8 range in data table) due to alkyl group on ester
[1 mark]

