| Date | May 2011 | Marks available | 1 | Reference code | 11M.1.hl.TZ1.20 |
| Level | HL | Paper | 1 | Time zone | TZ1 |
| Command term | Deduce | Question number | 20 | Adapted from | N/A |
Question
Curve X on the graph below shows the volume of oxygen formed during the catalytic decomposition of a \({\text{1.0 mol}}\,{\text{d}}{{\text{m}}^{ - 3}}\) solution of hydrogen peroxide.
\[{\text{2}}{{\text{H}}_2}{{\text{O}}_2}{\text{(aq)}} \to {{\text{O}}_2}{\text{(g)}} + {\text{2}}{{\text{H}}_2}{\text{O(l)}}\]
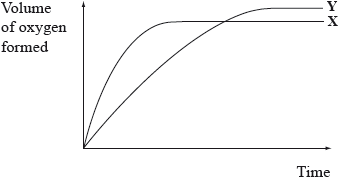
Which change would produce the curve Y?
A. Adding water
B. Adding some 0.1 mol dm–3 hydrogen peroxide solution
C. Using a different catalyst
D. Lowering the temperature
Markscheme
B
Examiners report
Two respondents stated that this question was somewhat misleading. The question was one of the more challenging questions on the paper but 51.70% of candidates did get B. as the correct answer.
Syllabus sections
Show 154 related questions
- 17N.3.sl.TZ0.2b: Suggest two variables, besides the time of reaction, which the student should have controlled...
- 17N.2.sl.TZ0.1e.ii: Suggest one possible reason for the differences between curves X and Y.
- 17N.2.sl.TZ0.1e.i: Explain the shape of curve X in terms of the collision theory.
- 17N.1.sl.TZ0.17: Excess magnesium powder was added to a beaker containing hydrochloric acid, HCl (aq). The...
- 17N.1.sl.TZ0.16: The diagram shows the energy profile for a catalysed and uncatalysed reaction.Which...
- 17M.2.sl.TZ1.1b: The student then carried out the experiment at other acid concentrations with all...
- 17M.2.sl.TZ1.1a.ii: A student produced these results with [H+] = 0.15 mol\(\,\)dm−3. Propanone and acid were in...
- 17M.2.sl.TZ1.1a.i: Suggest how the change of iodine concentration could be followed.
- 17M.1.sl.TZ1.17: 100 cm3 of 10% hydrogen peroxide solution decomposes at 298 K to form water and...
- 17M.1.sl.TZ1.16: Copper catalyses the reaction between zinc and dilute sulfuric acid. Zn(s) + H2SO4(aq) →...
- 16N.2.sl.TZ0.3g: Suggest one reason why the values of rates of reactions obtained at higher temperatures may...
- 16N.2.sl.TZ0.3f: An additional experiment was carried out at a higher temperature, T2. (i) On the same axes,...
- 16N.2.sl.TZ0.3e: A student decided to carry out another experiment using 0.075 mol dm-3 solution of sodium...
- 16N.2.sl.TZ0.3d: Draw the best fit line of \(\frac{1}{{\rm{t}}}\) against concentration of sodium thiosulfate...
- 16N.1.sl.TZ0.17: Which experimental methods could be used to observe the progress of the following...
- 16N.1.sl.TZ0.16: For the reaction R → P, which letter represents the activation energy for the catalysed...
- 16M.2.hl.TZ0.3b: The enthalpy change for the reaction between nitrogen monoxide and hydrogen is −664 kJ and...
- 16M.2.hl.TZ0.3a: (i) State the equation for the overall reaction. (ii) Deduce the rate expression consistent...
- 16M.2.sl.TZ0.3b: (i) Sketch the potential energy profile for the synthesis of phosgene, using the axes given,...
- 16M.1.sl.TZ0.17: Graph 1 shows a plot of volume...
- 16M.1.sl.TZ0.16: Which conditions must be met for a reaction to take place? I. Reactants collide with...
- 15M.2.hl.TZ1.3a: Define the term rate of reaction.
- 15M.2.hl.TZ1.3b: Describe the collision theory.
- 15M.2.hl.TZ2.2a: Define the term rate of reaction.
- 15M.2.hl.TZ2.2b: Explain why increasing the particle size of a solid reactant decreases the rate of reaction.
- 15M.2.hl.TZ2.7a.iii: Define the term activation energy, \({E_{\text{a}}}\).
- 15M.2.hl.TZ2.7b: Ammonia is manufactured by the Haber process in which iron is used as a catalyst. Explain...
- 15M.1.sl.TZ1.18: Nitrogen gas reacts with hydrogen gas according to the following...
- 15M.1.sl.TZ1.17: Which variable is best to use when determining the rate of decomposition of hydrogen...
- 15M.1.sl.TZ2.17: Which is a correct unit for expressing the rate of a reaction? A. ...
- 15M.1.sl.TZ2.18: \({\text{100 c}}{{\text{m}}^{\text{3}}}\) of a...
- 15M.2.sl.TZ1.3a: Define the term rate of reaction.
- 15M.2.sl.TZ1.3b: Describe the collision theory.
- 15M.2.sl.TZ2.5d.ii: Ammonia is manufactured by the Haber process in which iron is used as a catalyst. Explain the...
- 15M.2.sl.TZ2.5d.iii: Sketch the Maxwell–Boltzmann energy distribution curve for a reaction, labelling both axes...
- 15M.2.sl.TZ1.7d.ii: Describe how a catalyst increases the rate of a reaction.
- 15M.2.sl.TZ2.5d.i: Define the term activation energy, \({E_{\text{a}}}\).
- 14M.1.hl.TZ2.22: Which pair of graphs shows a decomposition reaction of \(X\) that obeys first-order kinetics?
- 14M.1.hl.TZ2.8: Which statements explain why a catalyst is used in the Contact process (shown...
- 14M.2.hl.TZ1.2a: Deduce, with a reason, the order of reaction with respect to each reactant.
- 14M.2.hl.TZ2.1d: Outline how you could establish that the system had reached equilibrium at the end of one week.
- 14M.2.hl.TZ2.1e: Outline why changing the temperature has only a very small effect on the value of the...
- 14M.2.hl.TZ2.6a: (i) State the volumes of the liquids that should be mixed. (ii) State why it is...
- 14M.2.hl.TZ2.6c: (i) Sketch and label, indicating an approximate activation energy, the Maxwell–Boltzmann...
- 14M.1.sl.TZ1.17: Why does the rate of a reaction increase when the temperature is increased? I. The...
- 14M.1.sl.TZ2.18: Which is not affected by an increase in temperature? A. Rate of reaction B. ...
- 14M.2.sl.TZ1.1a: (i) The graph shows the volume of hydrogen produced against time under these experimental...
- 14M.1.sl.TZ1.18: The diagram represents the Maxwell‒Boltzmann energy distribution curve of the reactants for a...
- 14M.1.sl.TZ2.17: Which change increases the rate of a chemical reaction? A. Increasing the size of solid...
- 14M.2.sl.TZ2.1b: The hydrochloric acid does not appear in the balanced equation for the reaction. State its...
- 14M.2.sl.TZ2.1g: Outline how you could establish that the system had reached equilibrium at the end of one week.
- 14M.2.sl.TZ2.1h: Outline why changing the temperature has only a very small effect on the value of the...
- 14M.2.sl.TZ2.5b: (i) State the volumes of the liquids that should be mixed. (ii) State why it is...
- 14M.2.sl.TZ2.5c: (i) Sketch and label, indicating an approximate activation energy, the Maxwell–Boltzmann...
- 14M.2.sl.TZ2.5e: (i) Calculate the volume of sulfur dioxide, in \({\text{c}}{{\text{m}}^{\text{3}}}\),...
- 14N.2.hl.TZ0.11a.ii: Explain how and why the rate of reaction changes with time.
- 14N.2.hl.TZ0.11b: A Maxwell-Boltzmann energy distribution curve is drawn below. Label both axes and explain, by...
- 14N.1.sl.TZ0.17: Consider the following reaction between hydrogen peroxide, hydrogen ions and iodide...
- 14N.1.sl.TZ0.18: Which quantity can be changed by the use of a catalyst? A. I and II only B. I and...
- 14N.2.sl.TZ0.5b: The Contact process operates at a temperature of 450 °C and a pressure of 2 atm as optimum...
- 14N.2.sl.TZ0.8e: (i) The experiment is repeated with the same amount of a more effective catalyst,...
- 13N.2.hl.TZ0.1a: The concentration of iodide ions, \({{\text{I}}^ - }\), is assumed to be constant. Outline...
- 13N.1.hl.TZ0.20: The diagram below shows the energy changes for a reaction with and without a catalyst. Which...
- 13N.2.hl.TZ0.1b: For this mixture the concentration of hydrogen peroxide,...
- 13N.2.hl.TZ0.1e: The colour change occurs when \(1.00 \times {10^{ - 4}}{\text{ mol}}\) of iodine has been...
- 13N.2.hl.TZ0.1g: In another experiment, 0.100 g of a black powder was also added while all other...
- 13N.1.sl.TZ0.18: The diagram below shows the energy changes for a reaction with and without a catalyst. Which...
- 13N.2.sl.TZ0.1j: In a third experiment, 0.100 g of a black powder was also added while all other...
- 13N.1.sl.TZ0.17: Which factors can increase the rate of a chemical reaction? I. Increasing the pressure...
- 13N.2.sl.TZ0.1d: For this mixture the concentration of hydrogen peroxide,...
- 13N.2.sl.TZ0.1i: In a second experiment, the concentration of the hydrogen peroxide was decreased to...
- 13N.2.sl.TZ0.1c: The concentration of iodide ions, \({{\text{I}}^ - }\), is assumed to be constant. Outline...
- 13N.2.sl.TZ0.1h: The colour change occurs when \(1.00 \times {10^{ - 4}}{\text{ mol}}\) of iodine has been...
- 13N.2.sl.TZ0.1k: Explain why increasing the temperature also decreases the time required for the colour to...
- 13M.2.hl.TZ1.3a: Define the term activation energy, \({E_{\text{a}}}\).
- 13M.1.sl.TZ1.18: Which statement best describes and explains the effect of a catalyst on the rate of a...
- 13M.1.sl.TZ1.17: Which graph best represents the relationship between the average kinetic energy of molecules...
- 13M.2.sl.TZ1.8c.i: Deduce the effect of the concentration of...
- 13M.2.sl.TZ1.8c.ii: Suggest why warm sodium hydroxide solution is used.
- 13M.2.hl.TZ2.2a: A catalyst provides an alternative pathway for a reaction, lowering the activation energy,...
- 13M.1.sl.TZ2.18: Which statements explain the increase in the rate of a reaction when the temperature is...
- 13M.2.sl.TZ2.2b: A catalyst provides an alternative pathway for a reaction, lowering the activation energy,...
- 13M.2.sl.TZ2.2c: Sketch two Maxwell–Boltzmann energy distribution curves for a fixed amount of gas at two...
- 12N.2.hl.TZ0.6a: (i) Define the term rate of reaction. (ii) Temperature and the addition of a...
- 12N.2.hl.TZ0.6f.ii: Deduce the rate expression for each step. Step 1: Step 2:
- 12N.2.hl.TZ0.6b: (i) Define the term activation energy, \({E_{\text{a}}}\). (ii) Sketch the two...
- 12N.2.hl.TZ0.6e.ii: Describe qualitatively the relationship between the rate constant, k, and temperature, T.
- 12N.1.sl.TZ0.17: Which piece of equipment could not be used in an experiment to measure the rate of this...
- 12N.2.sl.TZ0.3c: (i) Define the term activation energy, \({E_{\text{a}}}\). (ii) Sketch the two...
- 12N.1.sl.TZ0.18: In which flask will the reaction between 2.0 g of magnesium carbonate and 25 cm3 1.0 mol...
- 10N.2.hl.TZ0.6a: (i) Use the graph to deduce whether the forward reaction is exothermic or endothermic and...
- 10N.3.hl.TZ0.E4: (a) (i) Explain the dependence of the dissociation of diatomic oxygen, O2, and ozone,...
- 10N.1.sl.TZ0.18: Which changes increase the rate of the reaction...
- 10N.1.sl.TZ0.17: A piece of zinc was added to aqueous nitric acid and the volume of hydrogen gas produced was...
- 10N.2.sl.TZ0.6a: (i) Use the graph to deduce whether the forward reaction is exothermic or endothermic and...
- 09N.2.hl.TZ0.6e.v: Suggest, stating a reason, how the addition of a catalyst at constant pressure and...
- 09N.2.hl.TZ0.6a.ii: Predict the effect on the rate of the forward reaction and on the rate constant if the...
- 09N.2.hl.TZ0.6c: State two situations when the rate of a chemical reaction is equal to the rate constant.
- 09N.1.sl.TZ0.18: Hydrochloric acid is reacted with large pieces of calcium carbonate, the reaction is then...
- 09N.1.sl.TZ0.19: Which statement is true about using sulfuric acid as a catalyst in the following...
- 09N.2.sl.TZ0.5c.i: The concentration of \({{\text{H}}_{\text{2}}}{{\text{O}}_{\text{2}}}\) is increased at...
- 09N.2.sl.TZ0.5a.iv: Suggest, stating a reason, how the addition of a catalyst at constant pressure and...
- 09N.2.sl.TZ0.5b: Graphing is an important method in the study of the rates of chemical reaction. Sketch a...
- 09N.2.sl.TZ0.5c.ii: The solution of NaI is prepared from a fine powder instead of large crystals.
- 09N.2.sl.TZ0.5d: Explain why the rate of a reaction increases when the temperature of the system increases.
- 17M.2.sl.TZ2.5a.ii: Sketch, on the same graph, the expected results if the experiment were repeated using...
- 17M.2.sl.TZ2.5a.i: Outline why the rate of the reaction decreases with time.
- 17M.1.sl.TZ2.17: Which methods can be used to monitor the progress of this...
- 17M.1.sl.TZ2.16: Which change does not increase the initial rate of reaction when CaCO3(s) is added to excess...
- 17M.2.hl.TZ1.1b: The student then carried out the experiment at other acid concentrations with all...
- 10M.2.hl.TZ1.2a: Explain why they added water to the mixtures.
- 10M.2.hl.TZ1.2d: (i) This reaction uses a catalyst. Sketch and annotate the Maxwell-Boltzmann energy...
- 10M.2.sl.TZ1.2: (a) Discuss whether either Alex’s or Hannah’s hypothesis is correct. (b) Explain why...
- 10M.2.sl.TZ2.6c: (i) Explain the shape of the curve. (ii) Copy the above graph on your answer sheet...
- 10M.1.sl.TZ2.18: Which unit could be used for the rate of a chemical reaction? A. mol B. ...
- 10M.1.sl.TZ2.19: Which of the following can increase the rate of a chemical reaction? I. Increasing the...
- 09M.1.sl.TZ1.18: What is the best definition of rate of reaction? A. The time it takes to use up all the...
- 09M.1.sl.TZ1.19: Which factors can affect reaction rate? I. The state of the reactants II. The...
- 09M.1.sl.TZ1.20: Equal masses of powdered calcium carbonate were added to separate solutions of hydrochloric...
- 09M.2.sl.TZ1.1d: The reactants had to be stirred vigorously because they formed two distinct layers in the...
- 09M.2.sl.TZ1.1c.iv: State and explain the effect that the addition of the sodium hydroxide catalyst will have on...
- 09M.2.hl.TZ2.7a.iii: The rate of this reaction in (a) (ii), can be studied by measuring the volume of gas...
- 09M.2.hl.TZ2.7a.i: Define the term rate of reaction.
- 09M.2.hl.TZ2.7a.iv: The experiment is repeated using a sample of hydrochloric acid with double the volume, but...
- 09M.2.hl.TZ2.7d.ii: Construct the enthalpy level diagram and label the activation energy, \({E_{\text{a}}}\), the...
- 09M.2.hl.TZ2.7d.iii: Describe qualitatively the relationship between the rate constant, \(k\), and the...
- 09M.2.hl.TZ2.7d.i: Define the term activation energy, \({E_{\text{a}}}\).
- 09M.1.sl.TZ2.17: Consider the reaction between magnesium and hydrochloric acid. Which factors will affect the...
- 09M.2.sl.TZ2.5a.iv: State and explain the effect of a catalyst on the position of equilibrium.
- 09M.2.sl.TZ2.4a: Define the term activation energy, \({E_{\text{a}}}\).
- 09M.2.sl.TZ2.4b: State two conditions necessary for a reaction to take place between two reactant particles.
- 09M.2.sl.TZ2.5a.iii: State the effect of a catalyst on the value of \({K_{\text{c}}}\).
- 11M.1.sl.TZ1.18: Consider the reaction between gaseous iodine and gaseous...
- 11M.1.sl.TZ1.17: Which statements describe the action of a catalyst? I. It does not alter the...
- 11M.2.sl.TZ1.3d: A catalyst of copper mixed with zinc oxide and alumina is used in industry for this...
- 11M.1.hl.TZ2.20: Sodium carbonate and hydrochloric acid react according to the equation...
- 11M.1.sl.TZ2.19: The reaction below represents the Haber process for the industrial production of...
- 11M.2.sl.TZ2.6a.v: The reaction can be catalysed by adding platinum metal. State and explain what effect the...
- 11M.2.sl.TZ2.7a.ii: List the three characteristic properties of reactant particles which affect the rate of...
- 11M.2.sl.TZ2.7a.iv: Explain why coal dust burns much faster than a large piece of coal with the same mass.
- 11M.2.sl.TZ2.7a.iii: On the axes below sketch two Maxwell-Boltzmann energy distribution curves for the same sample...
- 11M.2.sl.TZ2.7a.i: Define the term rate of a chemical reaction.
- 12M.1.sl.TZ2.18: The following enthalpy level diagram shows the effect of the addition of a catalyst on a...
- 12M.1.sl.TZ2.17: Which are appropriate units for the rate of a reaction? A. ...
- 12M.2.sl.TZ2.1c: (i) Draw a curve on the graph opposite to show how the height of the bubble layer changes...
- 12M.2.sl.TZ2.1a: Explain why the curve reaches a maximum.
- 12M.2.sl.TZ2.1b: Use the graph to calculate the rate of decomposition of hydrogen peroxide at 120 s.
- 11N.1.sl.TZ0.17: A student added 0.20 g of calcium carbonate powder to...
- 11N.1.sl.TZ0.18: Which statement about the kinetic theory is not correct? A. The particles in ice vibrate...
- 11N.2.sl.TZ0.1d.i: It takes just 0.0400 seconds to produce nitrogen gas in the simulation. Calculate the average...
- 11N.2.sl.TZ0.1d.ii: The students also discovered that a small increase in temperature (e.g. 10 °C) causes a large...
- 11N.2.sl.TZ0.6e.i: Define the term rate of reaction.
- 11N.2.sl.TZ0.6b: Define the term activation energy, \({E_{\text{a}}}\).
- 11N.2.sl.TZ0.6d.ii: Sketch the Maxwell-Boltzmann energy distribution curve for a reaction with and without a...

