Topics 2.1 and 2.2
Paper 1 style questions are multiple choice. You are not permitted to use a calculator or the data book for these questions, but you should use a periodic table.
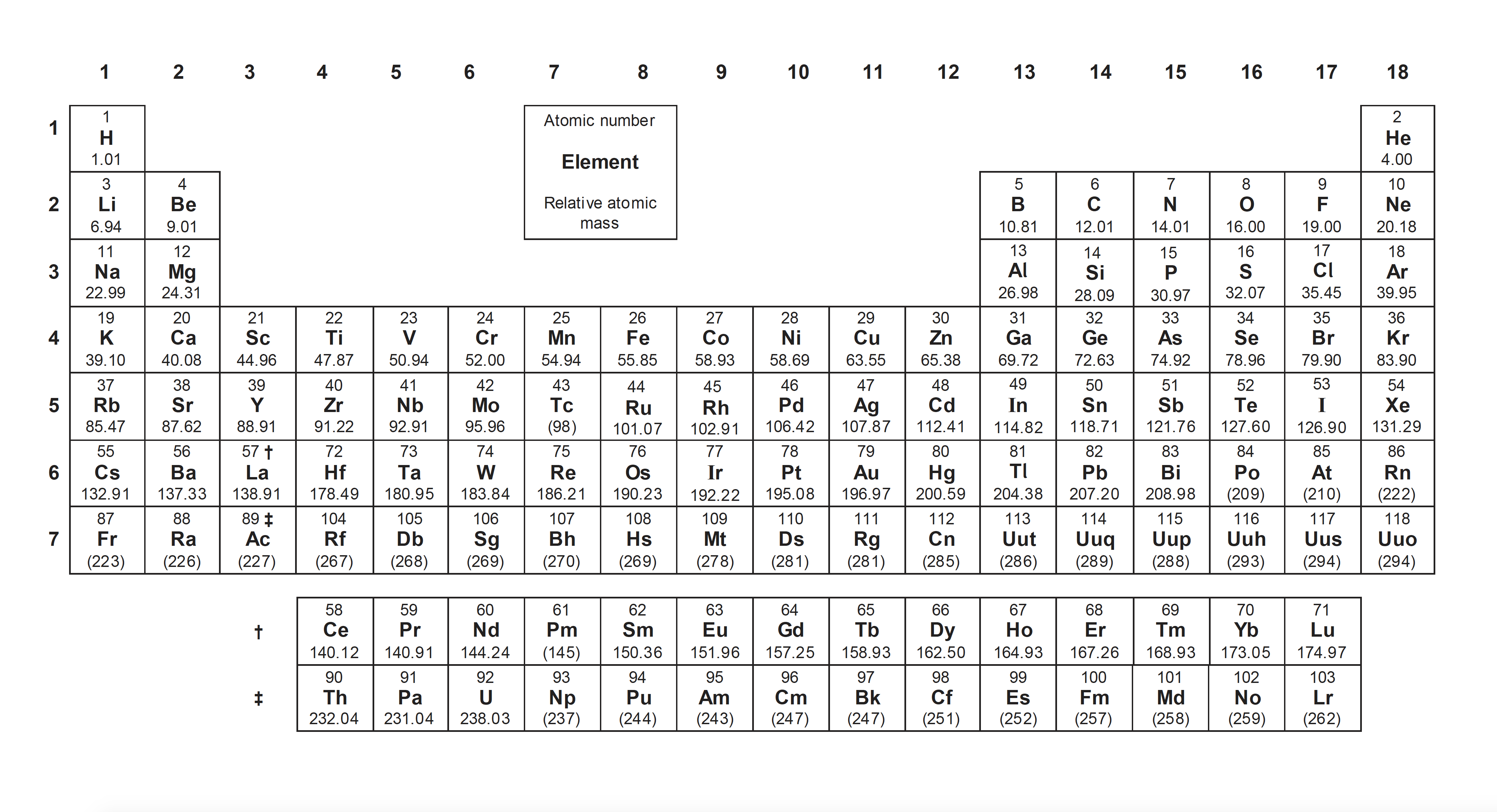
A periodic table pop-up is available on the left hand menu.
Quiz
Which of the following best represents the shape of a p-atomic orbital?
Orbitals are plots of electron position (90% probability) around the nucleus. They are three dimensional (not planar). An s-orbital is a sphere; a p-orbital is a three dimensional figure-of-eight.
So the answer here that best decribes a p-orbital is  .
.
The blue spherical image represents an s-orbital.
For HL students only: The elongated blue image represents a sigma molecular orbital (two orbitals overlapping end on) and the elongated red image represents a pi molecular orbital (two p-orbitals overlapping side on).
What is the correct electronic configuration for an atom of chromium?
The sub-levels of an atom are filled in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s. Remember that 's' sub-levels hold a maximum of 2 electrons, 'p' hold 6, and 'd' hold 10. A chromium atom has 24 electrons in total (atomic number is 24), but the configuration is not what would be expected as the six electrons in the 4s and 3d energy levels are all in singly-occupied orbitals (4s1 3d5) instead of filling the sub-levels in the conventional manner (4s2 3d4). Chromium and copper have unusual configurations and need to be learned.
Remember that [Ar] is shorthand and indicates the electron configuration of argon: 1s22s22p63s23p6.
The correct answer is therefore 1s2 2s22p63s23p64s13d5
Which is the correct number of protons, neutrons and electrons in the selenide ion \(\begin{smallmatrix} 79\\ 34 \end{smallmatrix}\)Se2−?
The upper number is the mass number, which is equal to protons plus neutrons. The lower number is the atomic number which is the number of protons. The number of electrons is equal to the number of protons in a neutral atom, but this is 2− negative ion, so it has two more electrons than protons.
So the species has 34 protons, (79−34=) 45 neutrons and 36 electrons (two more than protons).
Which statements are correct with regard to the emission spectrum of hydrogen?
1: The lines are produced when electrons move from higher to lower energy levels
2: The lines converge at lower frequencies
3: The transitions to n=1 are seen in the visible region of the electromagnetic spectrum
The line emission spectrum of hydrogen is composed of discrete lines that represent particular frequencies of light. Lines are produced by electron transitions from higher to lower energy levels (for emission; from lower to higher for absorption).
The lines converge at higher frequencies (not lower).
Electron transitions in the hydrogen emission spectrum from higher energy levels to n=1 result in emissions in the ultra-violet region (highest energy since these transitions have the largest gaps between energy levels). Transitions to n=2 are found in the visible region. Transitions to n=3 are found in the infra-red region (lowest energy since these transitions have the smallest gaps between energy levels).
Thus the answer is 1 only.
Which statement about 51V2+ and 50V3+ is correct?
The upper (superscript) number on the left is the mass number, which is equal to protons plus neutrons. The atomic number for vanadium is 23, which is the number of protons; shown on the periodic table. All atoms of vanadium have the same number of protons. 51V2+ therefore has 51−23=28 neutrons and 50V3+ has 50−23=27 neutrons.
The number of electrons is equal to the number of protons in a neutral atom, but these species are positive ions so will have more protons than electrons.
The correct answer is therefore Both have the same number of protons.
In which set do all the species contain more electrons than neutrons?
The upper (superscript) number on the left is the mass number, which is equal to protons plus neutrons. The atomic number, which is the number of protons is shown on the periodic table. The number of electrons is equal to the number of protons in a neutral atom but negative ions will have correspondingly more electrons:
32S2− has 32−16=16 neutrons and 16+2=18 electrons
32S has 32−16=16 neutrons and 16 electrons
31P3− has 31−15=16 neutrons and 15+3=18 electrons
31P has 31−15=16 neutrons and 15 electrons
13N3− has 13−7=6 neutrons and 7+3=10 electrons
13N has 13−7=6 neutrons and 7 electrons
The correct answer is therefore 32S2−, 31P3−, 13N as this is the only set in which all species contain more electrons than neutrons.
Which electron transition in the hydrogen absorption spectrum absorbs radiation with the longest wavelength?
Longer wavelength corresponds to lower frequency/lower energy. So the longest wavelength will correspond to the electron transition of lowest energy.
The question also asks about absorption - energy taken in - so the transition must be from a lower to higher energy level.
The energy gaps between the levels gets smaller at higher energy.
Therefore the smallest energy transition that is also an absorption is n=3 to n=4, which is therefore the correct answer.
The full electron configuration of an atom of an element is 1s2 2s2 2p6 3s2 3p4.
To which group and period does the element belong?
The electron configuration reflects an element's position on the periodic table.
The easiest way to solve this is to count the total number of electrons (16); as this is an atom, the number of electrons will be equal to the number of protons, so the element is sulfur.
Sulfur is in Group 16, Period 3.
Which statement is correct for the fluoride ion \(\begin{smallmatrix} 19\\ 9 \end{smallmatrix}\)F−?
The upper number is the mass number, which is equal to protons plus neutrons (19 sub-atomic particles in the nucleus). The lower number is the atomic number which is the number of protons. The number of electrons is equal to the number of protons in a neutral atom, but this is a 1− negative ion, so it has one more electron than protons.
So the species has 9 protons, (19−9=)10 neutrons and 10 electrons (1s2 2s2 2p6).
The correct answer is therefore Most of the volume of the ion is unoccupied 'space' which is of course the case with all atoms and ions, since most of the mass is concentrated in the nucleus.
What is the number of protons and the number of neutrons in 89Rb?
The upper number on the left is the mass number, which is equal to protons plus neutrons. The atomic number for rubidium is 37, which is the number of protons; shown on the periodic table.
89Rb therefore has 37 protons and (89−37=)52 neutrons.
The correct answer is therefore 37 protons, 52 neutrons.
What is the correct condensed electronic configuration for the Co2+ ion?
The sub-levels of an atom are filled in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s.
Remember that 's' sub-levels hold a maximum of 2 electrons, 'p' hold 6, and 'd' hold 10, and that [Ar] is shorthand and indicates the electron configuration of argon: 1s22s22p63s23p6.
A cobalt atom has 27 electrons in total (atomic number is 27) so the condensed atomic electron configuration is [Ar] 4s2 3d7.
The Co2+ ion has a 2+ charge so has lost two electrons. These electrons are lost from the 4s orbital before the 3d. Remember that '4s are gained first and lost first'!
The correct answer is therefore [Ar] 4s0 3d7.
What is the maximum number of electrons that can occupy the n=4 energy level?
Each and every orbital can hold two electrons. Any s sub-level contains one orbital and can therefore hold two electrons. Any p sub-level contains three orbitals and can therefore hold six electrons. Any d sub-level contains five orbitals and can therefore hold ten electrons. Any f sub-level contains seven orbitals and can therefore hold fourteen electrons.
The n=4 energy level consists of the 4s, 4p, 4d and 4f sub-levels.
Thus the answer is 32 (2+6+10+14).
Paper 1 style questions are multiple choice. You are not permitted to use a calculator or the data book for these questions, but you may use a periodic table.
Quiz
Which of the following best represents the shape of an s-atomic orbital?
Orbitals are plots of electron position (90% probability) around the nucleus. They are three dimensional (not planar). An s-orbital is a sphere; a p-orbital is a three dimensional figure-of-eight.
So the answer here that best decribes as s-orbital is  .
.
The red figure-of-eight image represents a p-orbital.
For HL students only: The elongated blue image represents a sigma molecular orbital (two orbitals overlapping end on) and the elongated red image represents a pi molecular orbital (two p-orbitals overlapping side on).
What is the correct electronic configuration for an atom of copper?
The sub-levels of an atom are filled in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s. Remember that 's' sub-levels hold a maximum of 2 electrons, 'p' hold 6, and 'd' hold 10.
A copper atom has 29 electrons in total (atomic number is 29), but the configuration is not what would be expected as the electrons in the 4s and 3d energy levels are 4s1 3d10 instead of filling the sub-levels in the conventional manner (4s2 3d9). Chromium and copper have unusual configurations and need to be learned.
Remember that [Ar] is shorthand and indicates the electron configuration of argon: 1s22s22p63s23p6.
The correct answer is therefore 1s2 2s22p63s23p64s13d10
Which is the correct number of protons, neutrons and electrons in the calcium ion \(\begin{smallmatrix} 44\\ 20 \end{smallmatrix}\)Ca2+?
The upper number is the mass number, which is equal to protons plus neutrons. The lower number is the atomic number which is the number of protons. The number of electrons is equal to the number of protons in a neutral atom, but this is 2+ positive ion, so it has two fewer electrons than protons.
So the species has 20 protons, (44−20) 24 neutrons and 18 electrons (two fewer than protons).
Which statements are correct with regard to the absorption spectrum of hydrogen?
1: The lines are produced when electrons move from higher to lower energy levels
2: The lines converge at lower wavelengths
3: The lines in the visible region of the electromagnetic spectrum are seen as coloured lines
The line absorption spectrum of hydrogen is composed of discrete lines that represent particular frequencies. Lines are produced by electron transitions from lower to higher energy levels (for absorption; from higher to lower for emission).
The lines converge at higher frequencies/energies which corresponds to lower wavelengths.
Any lines in the absorption spectrum are not seen as coloured lines, but as black lines (an absence of colour) as electromagnetic radiation is absorbed (not emitted).
Thus the answer is 2 only.
Which statement about 65Zn2+ and 65Cu+ is correct?
The upper number on the left is the mass number, which is equal to protons plus neutrons. The atomic number for zinc is 30 and for copper is 29; this is the number of protons, shown on the periodic table.
The number of electrons is equal to the number of protons in a neutral atom, but these species are positive ions so will have more protons than electrons.
65Zn2+ therefore has 30 protons, 65−30=35 neutrons, and 30−2=28 electrons (as it is a 2+ ion).
65Cu+ therefore has 29 protons, 65−29=36 neutrons, and 29−1=28 electrons (as it is a 1+ ion).
The correct answer is therefore Both have the same number of electrons.
In which set do all the species contain more neutrons than electrons?
The upper (superscript) number on the left is the mass number, which is equal to protons plus neutrons. The atomic number, which is the number of protons is shown on the periodic table. The number of electrons is equal to the number of protons in a neutral atom but negative ions will have correspondingly more electrons and positive ions correspondingly fewer.
8Li+ has 8−3=5 neutrons and 3−1=2 electrons
8Li has 8−3=5 neutrons and 3 electrons
31P3− has 31−15=16 neutrons and 15+3=18 electrons
31P has 31−15=16 neutrons and 15 electrons
15N3− has 15−7=8 neutrons and 7+3=10 electrons
15N has 15−7=8 neutrons and 7 electrons
The correct answer is therefore 8Li+, 31P, 15N as this is the only set in which all species contain more neutrons than electrons.
Which electron transition in the hydrogen emission spectrum emits radiation with the longest wavelength?
Longer wavelength corresponds to lower frequency/lower energy. So the longest wavelength will correspond to the electron transition of lowest energy.
The question also asks about emission - energy given out - so the transition must be from a higher to lower energy level.
The energy gaps between the levels get smaller at higher energy.
Therefore the smallest energy transition that is also an emission is n=6 to n=5, which is therefore the correct answer.
The full electron configuration of an atom of an element is 1s2 2s2 2p6 3s2 3p6 4s2 3d3.
To which group and period does the element belong?
The electron configuration reflects an element's position on the periodic table.
The easiest way to solve this is to count the total number of electrons (23); as this is an atom, the number of electrons will be equal to the number of protons, so the element is vanadium.
Vanadium is in Group 5, Period 4.
Fluorine has only one stable isotope. Which statement is incorrect for the fluoride ion \(\begin{smallmatrix} 19\\ 9 \end{smallmatrix}\)F−?
The upper number is the mass number, which is equal to protons plus neutrons (19 sub-atomic particles in the nucleus). The lower number is the atomic number which is the number of protons. The number of electrons is equal to the number of protons in a neutral atom, but this is a 1− negative ion, so it has one more electron than protons.
So this fluoride ion has 9 protons, (19−9=)10 neutrons and 10 electrons (1s2 2s2 2p6).
The question states that there is only one stable isotope of fluorine so all stable fluoride ions will have 10 neutrons.
The correct answer is therefore The ion has the same electron configuration as an atom of argon as this is incorrect: Argon has an electron configuration of 1s2 2s2 2p6 3s2 3p6.
What is the number of protons and the number of neutrons in 82Kr?
The upper number on the left is the mass number, which is equal to protons plus neutrons. The atomic number for krypton is 36, which is the number of protons; shown on the periodic table.
82Kr therefore has 36 protons and (82−36=)46 neutrons.
The correct answer is therefore 36 protons, 46 neutrons.
What is the correct condensed electronic configuration for the V3+ ion?
The sub-levels of an atom are filled in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s.
Remember that 's' sub-levels hold a maximum of 2 electrons, 'p' hold 6, and 'd' hold 10, and that [Ar] is shorthand and indicates the electron configuration of argon: 1s22s22p63s23p6.
A vanadium atom has 23 electrons in total (atomic number is 23) so the condensed atomic electron configuration is [Ar] 4s2 3d3.
The V3+ ion has a 3+ charge so has lost three electrons. These electrons are lost from the 4s orbital before the 3d. Remember that '4s are gained first and lost first'! So two electrons are lost from 4s and one electron is lost from 3d.
The correct answer is therefore [Ar] 4s0 3d2.
What is the maximum number of electrons that can occupy the n=3 energy level?
Each and every orbital can hold two electrons. Any s sub-level contains one orbital and can therefore hold two electrons. Any p sub-level contains three orbitals and can therefore hold six electrons. Any d sub-level contains five orbitals and can therefore hold ten electrons. Any f sub-level contains seven orbitals and can therefore hold fourteen electrons.
The n=3 energy level consists of the 3s, 3p, and 3d sub-levels.
Thus the answer is 18 (2+6+10).
How much of Atomic Structure core (SL and HL) paper 1 questions have you understood?


 Twitter
Twitter  Facebook
Facebook  LinkedIn
LinkedIn