| Date | November 2018 | Marks available | 3 | Reference code | 18N.2.hl.TZ0.6 |
| Level | HL | Paper | 2 | Time zone | TZ0 |
| Command term | Sketch | Question number | 6 | Adapted from | N/A |
Question
Butanoic acid, CH3CH2CH2COOH, is a weak acid and ethylamine, CH3CH2NH2, is a weak base.
State the equation for the reaction of each substance with water.
Draw a diagram showing the delocalization of electrons in the conjugate base of butanoic acid.
Deduce the average oxidation state of carbon in butanoic acid.
A 0.250 mol dm−3 aqueous solution of butanoic acid has a concentration of hydrogen ions, [H+], of 0.00192 mol dm−3. Calculate the concentration of hydroxide ions, [OH−], in the solution at 298 K.
Determine the pH of a 0.250 mol dm−3 aqueous solution of ethylamine at 298 K, using section 21 of the data booklet.
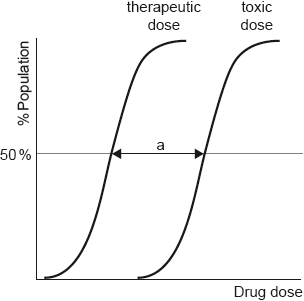
Sketch the pH curve for the titration of 25.0 cm3 of ethylamine aqueous solution with 50.0 cm3 of butanoic acid aqueous solution of equal concentration. No calculations are required.
Explain why butanoic acid is a liquid at room temperature while ethylamine is a gas at room temperature.
State a suitable reagent for the reduction of butanoic acid.
Deduce the product of the complete reduction reaction in (e)(i).
Markscheme
Butanoic acid:
CH3CH2CH2COOH (aq) + H2O (l) CH3CH2CH2COO− (aq) + H3O+ (aq) ✔
Ethylamine:
CH3CH2NH2 (aq) + H2O (l) CH3CH2NH3+ (aq) + OH− (aq) ✔
Diagram showing:
dotted line along O–C–O AND negative charge
Accept correct diagrams with pi clouds.
–1 ✔
«» = 5.21 × 10–12 «mol dm–3» ✔
«pKb = 3.35, Kb = 10–3.35 = 4.5 × 10–4»
«C2H5NH2 + H2O C2H5NH3+ + OH–»
Kb =
OR
«Kb =» 4.5 × 10–4 =
OR
«Kb =» 4.5 × 10–4 = ✔
« x = [OH–] =» 0.011 «mol dm–3» ✔
«pH = –log» 12.04
OR
«pH = 14.00 – (–log 0.011)=» 12.04 ✔
Award [3] for correct final answer.
decreasing pH curve ✔
pH close to 7 (6–8) at volume of 25 cm3 butanoic acid ✔
weak acid/base shape with no flat «strong acid/base» parts on the curve ✔
Any two of:
butanoic acid forms more/stronger hydrogen bonds ✔
butanoic acid forms stronger London/dispersion forces ✔
butanoic acid forms stronger dipole–dipole interaction/force ✔
Accept “butanoic acid forms dimers”
Accept “butanoic acid has larger Mr/hydrocarbon chain/number of electrons” for M2.
Accept “butanoic acid has larger «permanent» dipole/more polar” for M3.
lithium aluminium hydride/LiAlH4 ✔
butan-1-ol/1-butanol/CH3CH2CH2CH2OH ✔