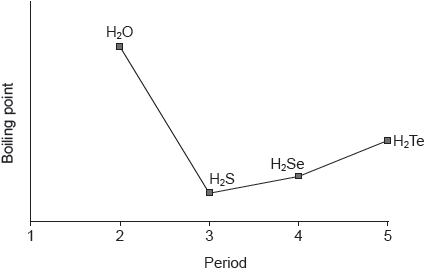
| Date | November 2016 | Marks available | 2 | Reference code | 16N.3.sl.TZ0.6 |
| Level | SL | Paper | 3 | Time zone | TZ0 |
| Command term | Suggest | Question number | 6 | Adapted from | N/A |
Question
Polymers are made up of repeating monomer units which can be manipulated in various ways to give structures with desired properties.
(i) Draw the structure of 2-methylpropene.
(ii) Deduce the repeating unit of poly(2-methylpropene).
Deduce the percentage atom economy for polymerization of 2-methylpropene.
(i) Suggest why incomplete combustion of plastic, such as polyvinyl chloride, is common in industrial and house fires.
(ii) Phthalate plasticizers such as DEHP, shown below, are frequently used in polyvinyl chloride.
With reference to bonding, suggest a reason why many adults have measurable levels of phthalates in their bodies.
Markscheme
i
OR
H2C=C(CH3)2
ii
OR
−CH2C(CH3)2−
Continuation bonds needed for mark.
No penalty if square brackets present or “n” appears after the bracket/formula.
«same mass of product as reactant, thus» 100«%»
Accept “less than 100%” only if a reason is given (eg, the catalyst is not converted into the product, or other reasonable answer).
i
due to stability of plastics/strong covalent bonds
OR
low volatility preventing good mixing with oxygen «gas»
OR
lack of/insufficient oxygen
OR
plastics are often parts of devices with non-combustible components «which mechanically prevent the combustion of plastic components»
OR
PVC already partly oxidised «because some C–H bonds are replaced with C–Cl bonds», so it cannot produce enough heat for complete combustion
OR
many industrial/household materials contain additives that reduce their flammability/act as flame retardants
ii
weakly bound to the PVC/no covalent bonds to PVC/only London/dispersion/instantaneous induced dipole-induced dipole forces between DEHP and PVC AND leach/evaporate «from PVC» to atmosphere/food chain
OR
has low polarity/contains non-polar hydrocarbon chains AND fat-soluble/deposits in the fatty tissues
OR
has unusual structural fragments/is a xenobiotic/difficult to metabolise AND stays in the body for a long time