| Date | May 2013 | Marks available | 1 | Reference code | 13M.1.sl.TZ1.16 |
| Level | SL | Paper | 1 | Time zone | TZ1 |
| Command term | State | Question number | 16 | Adapted from | N/A |
Question
Which combination is correct about the energy changes during bond breaking and bond formation?
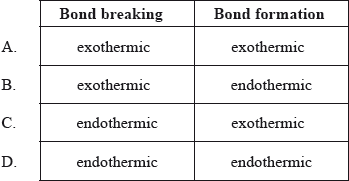
Markscheme
C
Examiners report
[N/A]
Syllabus sections
Show 64 related questions
- 17N.1.sl.TZ0.15: What is the enthalpy change, in kJ, of the following reaction? 3H2 (g) + N2 (g)...
- 17N.2.hl.TZ0.1e: Suggest why the enthalpy change of neutralization of CH3COOH is less negative than that of HCl.
- 17M.2.sl.TZ2.5b: Nitrogen dioxide and carbon monoxide react according to the following equation: NO2(g) +...
- 17M.1.sl.TZ2.15: What can be deduced from the facts that ozone absorbs UV radiation in the region of 340 nm...
- 17M.1.sl.TZ2.13: What can be deduced from this reaction profile? A. The reactants are less stable than...
- 17M.1.sl.TZ1.15: In which order does the oxygen–oxygen bond enthalpy increase? A. H2O2 < O2 <...
- 16N.3.sl.TZ0.6c: (i) Suggest why incomplete combustion of plastic, such as polyvinyl chloride, is common in...
- 16N.2.hl.TZ0.1b: (i) Calculate ΔHθ, in kJ, for this similar reaction below using \(\Delta H_{\rm{f}}^\theta \)...
- 16N.2.sl.TZ0.1a: Ethane-1,2-diol can be formed according to the following reaction. 2CO (g) +...
- 16N.1.sl.TZ0.14: In which reaction do the reactants have a lower potential energy than the products? A....
- 16N.1.sl.TZ0.10: The C=N bond has a bond length of 130 pm and an average bond enthalpy of 615kJmol-1. Which...
- 16M.2.hl.TZ0.3b: The enthalpy change for the reaction between nitrogen monoxide and hydrogen is −664 kJ and...
- 16M.2.sl.TZ0.3b: (i) Sketch the potential energy profile for the synthesis of phosgene, using the axes given,...
- 16M.1.sl.TZ0.15: Which equation represents the average bond...
- 15M.2.hl.TZ1.5b.i: Define the term average bond enthalpy.
- 15M.1.sl.TZ1.16: Which equation corresponds to the bond enthalpy of the H–I bond? A. ...
- 15M.1.sl.TZ2.16: Which enthalpy changes can be calculated using only bond enthalpy data? I. ...
- 15M.2.sl.TZ1.5b.i: Define the term average bond enthalpy.
- 15M.2.sl.TZ2.7g.i: Define the term average bond enthalpy.
- 15M.2.sl.TZ2.1b.ii: Calculate the enthalpy change, in \({\text{kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}\), for the...
- 14M.3.hl.TZ1.16d: Explain, with the aid of Lewis (electron-dot) structures, the difference between oxygen and...
- 14N.2.hl.TZ0.2c: Comment on which of the values obtained in (a) and (b)(ii) is more accurate, giving a reason.
- 13N.1.hl.TZ0.15: Which processes are exothermic? I. ...
- 13N.2.hl.TZ0.5c: Calculate the enthalpy change, in \({\text{kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}\), when one...
- 13N.1.sl.TZ0.15: Which processes are exothermic? I. ...
- 13N.2.sl.TZ0.4c: Calculate the enthalpy change, in \({\text{kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}\), when one...
- 13N.2.sl.TZ0.4g.iii: Predict, with an explanation, how the result obtained would compare with the value in Table...
- 13M.1.sl.TZ1.15: Which statement is correct for the enthalpy level diagram shown? A. The reaction is...
- 13M.1.sl.TZ2.17: The reaction between methane and oxygen is...
- 13M.2.sl.TZ2.6c.ii: Determine the enthalpy change, \(\Delta H\), in...
- 12N.2.sl.TZ0.2a: Define the term average bond enthalpy.
- 12N.2.sl.TZ0.2b: (i) Determine \(\Delta H\), the enthalpy change of the reaction, in...
- 10N.3.hl.TZ0.E4: (a) (i) Explain the dependence of the dissociation of diatomic oxygen, O2, and ozone,...
- 10N.1.sl.TZ0.16: Which equation best represents the bond enthalpy of HCl? A. ...
- 10N.2.sl.TZ0.4d: Hydrazine is a valuable rocket fuel. The equation for the reaction between hydrazine and...
- 10N.3.sl.TZ0.B1a: Glucose, \({{\text{C}}_{\text{6}}}{{\text{H}}_{{\text{12}}}}{{\text{O}}_{\text{6}}}\), is a...
- 09N.2.hl.TZ0.7b.iv: Using bond enthalpy values, calculate \(\Delta {H^\Theta }\) for the following...
- 09N.2.hl.TZ0.7b.v: Suggest with a reason, why the values obtained in parts (b) (i) and (b) (iv) are different.
- 09N.1.sl.TZ0.17: Use the average bond enthalpies below to calculate the enthalpy change, in kJ, for the...
- 10M.2.hl.TZ2.4e: Comment on the environmental safety of the products of the reaction of...
- 09M.2.hl.TZ1.3c: Suggest one reason why John’s answer is slightly less accurate than Marit’s answer and...
- 09M.2.hl.TZ1.3b: Determine the value for the enthalpy of hydrogenation of ethene using the values for the...
- 09M.1.sl.TZ1.16: Which process represents the C–Cl bond enthalpy in tetrachloromethane? A. ...
- 09M.2.sl.TZ1.2a: Calculate the value for the enthalpy of hydrogenation of ethene obtained using the average...
- 09M.2.sl.TZ1.2c: Suggest one reason why John’s answer is slightly less accurate than Marit’s answer.
- 09M.2.sl.TZ1.2d.i: Use the average bond enthalpies to deduce a value for the enthalpy of hydrogenation of...
- 09M.2.sl.TZ1.2d.ii: The percentage difference between these two methods (average bond enthalpies and enthalpies...
- 09M.2.hl.TZ2.4a: The standard enthalpy change of formation for...
- 09M.2.sl.TZ2.6a.ii: Use the information from Table 10 of the Data Booklet to determine the standard enthalpy...
- 09M.2.sl.TZ2.4c: Sketch an enthalpy level diagram to describe the effect of a catalyst on an exothermic reaction.
- 09M.2.sl.TZ2.6a.i: Define the term average bond enthalpy.
- 11M.1.sl.TZ1.15: Which statement about bonding is correct? A. Bond breaking is endothermic and requires...
- 11M.2.sl.TZ1.1a: Using the information from Table 10 of the Data Booklet, determine the theoretical enthalpy...
- 11M.2.sl.TZ1.1c.i: Part (a)
- 11M.1.hl.TZ2.17: Which equation represents the bond enthalpy for the H–Br bond in hydrogen bromide? A. ...
- 11M.2.sl.TZ2.6b.iv: Explain why the enthalpy of hydrogenation of propene is an exothermic process.
- 11M.2.sl.TZ2.6b.iii: Determine a value for the hydrogenation of propene using information from Table 10 of the...
- 11M.2.sl.TZ2.6b.ii: Enthalpy changes can be determined using average bond enthalpies. Define the term average...
- 12M.3.hl.TZ1.E1a: Oxygen absorbs much of the ultraviolet (UV) radiation from the sun, but ozone is important...
- 12M.3.sl.TZ1.B1a: (i) Calculate the heat required, in kJ, to raise the temperature of the water, using data...
- 12M.3.hl.TZ2.E3a: Explain, in terms of their bonding, how the presence of oxygen and ozone in the ozone layer...
- 11N.1.sl.TZ0.14: A student measured the temperature of a reaction mixture over time using a temperature probe....
- 11N.2.sl.TZ0.4a: Define the term average bond enthalpy.
- 11N.2.sl.TZ0.4c: Determine the standard enthalpy change, in \({\text{kJ}}\,{\text{mo}}{{\text{l}}^{ - 1}}\),...

