| Date | May 2019 | Marks available | 1 | Reference code | 19M.3.sl.TZ2.2 |
| Level | SL | Paper | 3 | Time zone | TZ2 |
| Command term | Examine | Question number | 2 | Adapted from | N/A |
Question
This question is about a mug made of a lead alloy.
The rate of lead dissolving in common beverages with various pH values was analysed.
Bromine and methanoic acid react in aqueous solution.
Br2 (aq) + HCOOH (aq) → 2Br− (aq) + 2H+ (aq) + CO2 (g)
The reaction was monitored by measuring the volume of carbon dioxide produced as time progressed.
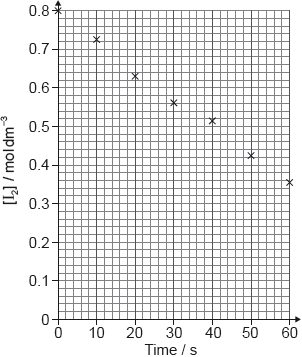
Determine from the graph the rate of reaction at 20 s, in cm3 s−1, showing your working.
Outline, with a reason, another property that could be monitored to measure the rate of this reaction.
Examine, giving a reason, whether the rate of lead dissolving increases with acidity at 18 °C.
Describe one systematic error associated with the use of the gas syringe, and how the error affects the calculated rate.
Identify one error associated with the use of an accurate stopwatch.
Markscheme
tangent drawn to curve at t = 20 s [✔]
slope/gradient calculation [✔]
0.35 «cm3 s–1» [✔]
Note: Accept values in the range 0.32–0.42 «cm3 s–1».
ALTERNATIVE 1
colour [✔]
Br2/reactant is coloured «Br– (aq)/product is not» [✔]
Note: Do not accept “changes in temperature” or “number of bubbles”.
ALTERNATIVE 2
conductivity [✔]
greater/increased concentration of ions in products [✔]
ALTERNATIVE 3
mass/pressure [✔]
gas is evolved/produced [✔]
Note: Do not accept “mass of products is less than mass of reactants”.
ALTERNATIVE 4
pH [✔]
methanoic acid is weak AND HBr is strong
OR
increase in [H+] [✔]
no AND experiment 7/beer has lowest rate and intermediate acidity/pH
OR
no AND experiment 6/orange juice has fastest rate but lower acidity/higher pH than experiment 5/lemonade
OR
no AND experiment 6/orange juice has highest rate and intermediate acidity/pH [✔]
Note: Accept no AND any comparison, with experimental support, that concludes no pattern/increase with acidity.
eg: “rate of Pb/lead dissolving generally decreases with acidity as tap water has highest rate (after orange juice) while lemonade (lower pH) has lower rate”.
ALTERNATIVE 1
gas may leak/be lost/escape
OR
plunger may stick/friction «so pressure is greater than atmospheric pressure»
OR
syringe may be tilted «up» so plunger moves less «with gravity acting on plunger»
OR
CO2 dissolved in water [✔]
calculated rate lower [✔]
ALTERNATIVE 2
syringe may be tilted «down» so plunger moves more «with gravity acting on plunger»
OR
syringe is held in hand so gets warmer and gas expands [✔]
calculated rate higher [✔]
Note: Calculated rate is lower or higher must be stated for M2.
Do not accept “scale on syringe is inaccurate”, “errors in reading syringe”, or “bubbles in syringe”.
human reaction time/delay «starting/stopping the stopwatch» [✔]
Note: Do not accept “inaccurate stopwatch”.
Examiners report
This question was challenging for many students. Quite a few candidates did draw a tangent line at 20s for 1 mark, show a slope/gradient calculation of the line for 1 mark, and had a reasonable final value for the final mark. Some candidates only found the average rate by finding the ratio of the value at that data point and received one mark (16/20=0.80 cm3 s-1). Candidates also received one mark if they had a correct answer with no work since the question clearly asked students to show their work.
The reaction rate was originally monitored by measuring the volume of CO2 produced. Students needed to propose another method for this reaction, with a reason, that could be used to measure the rate. There were several possible correct answers and most students received at least one mark with many receiving both marks. The most common incorrect answer involved changes in temperature.
This question required students to recognize the rate of lead dissolving did not increase with acidity and to refer to data in the table for the reason. Some students did not refer to data in the table and did not receive the mark because they did not have a reason, other students compared the rate of lead dissolving with temperature increasing which did not answer the question.
This question was asking about a systematic error. There were several possible correct answers for the error, but students also needed to clearly identify a specific error and if the rate increased or decreased for the second mark. Many students confused this with the concept of a random error and identified the uncertainty of reading the syringe which is incorrect. Teachers need to reinforce the concept of systematic versus random errors.
This question was well answered by most candidates although some students did not read the question clearly and commented on the stopwatch having problems or not being accurate.