| Date | May 2019 | Marks available | 2 | Reference code | 19M.2.sl.TZ2.2 |
| Level | SL | Paper | 2 | Time zone | TZ2 |
| Command term | Outline | Question number | 2 | Adapted from | N/A |
Question
The thermal decomposition of dinitrogen monoxide occurs according to the equation:
2N2O (g) → 2N2 (g) + O2 (g)
The reaction can be followed by measuring the change in total pressure, at constant temperature, with time.
The x-axis and y-axis are shown with arbitrary units.
Explain why, as the reaction proceeds, the pressure increases by the amount shown.
Outline, in terms of collision theory, how a decrease in pressure would affect the rate of reaction.
The experiment is repeated using the same amount of dinitrogen monoxide in the same apparatus, but at a lower temperature.
Sketch, on the axes in question 2, the graph that you would expect.
The experiment gave an error in the rate because the pressure gauge was inaccurate. Outline whether repeating the experiment, using the same apparatus, and averaging the results would reduce the error.
The graph below shows the Maxwell–Boltzmann distribution of molecular energies at a particular temperature.
The rate at which dinitrogen monoxide decomposes is significantly increased by a metal oxide catalyst.
Annotate and use the graph to outline why a catalyst has this effect.
Markscheme
increase in the amount/number of moles/molecules «of gas» [✔]
from 2 to 3/by 50 % [✔]
«rate of reaction decreases»
concentration/number of molecules in a given volume decreases
OR
more space between molecules [✔]
collision rate/frequency decreases
OR
fewer collisions per second/unit time [✔]
Note: Do not accept just “larger space/volume” for M1.
smaller initial gradient [✔]
initial pressure is lower AND final pressure of gas lower «by similar factor» [✔]
no AND it is a systematic error/not a random error
OR
no AND «a similar magnitude» error would occur every time [✔]
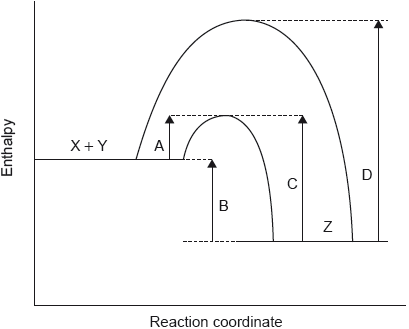
catalysed and uncatalysed Ea marked on graph AND with the catalysed being at lower energy [✔]
«for catalysed reaction» greater proportion of/more molecules have E ≥ Ea / E > Ea
OR
«for catalysed reaction» greater area under curve to the right of the Ea [✔]
Note: Accept “more molecules have the activation energy”.
Examiners report
About a quarter of the candidates gave the full answer. Some only gained the first marking point (M1) by recognizing the increase in the number of moles of gas. Some candidates wrote vague answers that did not receive credit such as “pressure increases as more gaseous products form” without explicitly recognizing that the reactants have fewer moles of gas than the products. Some candidates mistook it for a system at equilibrium when the pressure stops changing (although a straight arrow is shown in the equation). A teacher commented that the wording of the question was rather vague “not clear if question is asking about stoichiometry (i.e. how 200 & 300 connect to coefficients) or rates (i.e. explain graph shape)”. We did not see a discussion of the slope of the graph with time and most candidates understood the question as it was intended.
More than half of the candidates obtained the mark allocated for “less frequent collisions” at lower pressure, but only strong candidates explained that this was due to the lower concentration or increased spacing between molecules. Some candidates talked about a decrease in kinetic energy and they did not show a good understanding of collision theory. Some candidates lost M1 for stating “fewer collisions” without reference to time or probability.
This was a challenging question. Candidates usually obtained only one of the two marks allocated for the answer. Most of them scored the mark for a lower initial slope at low temperature, while others scored a mark for sketching their curve below the original curve as all pressures (initial and final) will be lower at the lower temperature. A teacher commented that the wording was unclear “sketch on the axes in question 2”, and it would have been better to label the graph instead.
This question was well answered by nearly 70 % of the candidates reflecting a good understanding of the impact of systematic errors. Some students did not gain the mark because of an incomplete answer. The question raised much debate among teachers. They worried if the error was clearly a systematic one. However, a high proportion of candidates had very clear and definite answers. In Spanish and French, the wording was a bit ambiguous which caused the markscheme in these languages to be more opened.
This question discriminated very well between high-scoring and low-scoring candidates. About half of the candidates annotated the Maxwell-Boltzmann distribution to show the effect of the catalyst. Some left it blank and some sketched a new distribution that would be obtained at a higher temperature instead. The majority of candidates knew that the catalyst provided an alternative route with lower Ea but only stronger candidates related it to the annotation of the graph and used the accurate language needed to score M2. A common mistake was stating that molecules have higher kinetic energy when a catalyst is added.