| Date | November 2016 | Marks available | 2 | Reference code | 16N.3.sl.TZ0.10 |
| Level | SL | Paper | 3 | Time zone | TZ0 |
| Command term | Identify and State | Question number | 10 | Adapted from | N/A |
Question
Amino acids are usually identified by their common names. Use section 33 of the data booklet.
State the IUPAC name for leucine.
A mixture of amino acids is separated by gel electrophoresis at pH 6.0. The amino acids are then stained with ninhydrin.
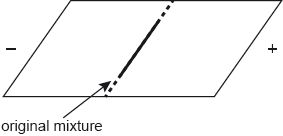
(i) On the diagram below draw the relative positions of the following amino acids at the end of the process: Val, Asp, Lys and Thr.
(ii) Suggest why glycine and isoleucine separate slightly at pH 6.5.
Determine the number of different tripeptides that can be made from twenty different amino acids.
The fibrous protein keratin has a secondary structure with a helical arrangement.
(i) State the type of interaction responsible for holding the protein in this arrangement.
(ii) Identify the functional groups responsible for these interactions.
Markscheme
2-amino-4-methylpentanoic acid
Accept 4-methyl-2-aminopentanoic acid.
i
Lys on cathode side AND Asp on anode side
Val at origin AND Thr on anode side but closer to origin than Asp
Val and Thr need not overlap.
Accept any (reasonable) size and demarcation of position so long as position relative to origin is correct.
Accept crosses for spots.
Award [1 max] for any two correct.
Award [1 max] if net direction of spots is reversed.
Award [1 max] if the four points are in the correct order but not in a straight line.
ii
different sizes/molar masses/chain lengths «so move with different speeds»
«203 =» 8000
i
hydrogen bonds
ii
carboxamide/amide/amido
OR
C=O AND N–H
Accept peptide.